 Jeroen van Duijneveldt's research group
Jeroen van Duijneveldt's research group
 Soft condensed matter
Soft condensed matter
The term soft condensed matter refers to a range of systems that fall between simple liquids and solids, for instance colloidal suspensions,
emulsions, liquid crystals, and polymers. This includes many systems of practical or biological importance, such as inks, paints, shampoos,
foodstuffs, milk and blood. Real systems tend to be complex, consisting of many components that are often difficult to characterise in detail.
Well-defined model systems are therefore studied instead. A central theme is the use of polymers to control particle interactions,
structure and phase behaviour in colloidal suspensions.
Please contact me if you are interested in studying for a Ph.D. in any of the areas listed
 Entropy driven gel formation and cluster phase
Entropy driven gel formation and cluster phase
For particles with hard interactions (closely approximated by colloids with steeply repulsive interactions), the internal energy does not depend on particle positions and any phase transitions are driven by an increase of entropy.
Rodlike particles can form liquid crystals and we have found this can lead to a
"nonsticky" gel state.
Thin discs can form a liquid crystalline (nematic) phase, but when curvature is introduced the nematic phase is frustrated and a transition to a locally ordered cluster phase is found. More commonly cluster phases arise from a competition between particle attractions and repulsions, but here the process is driven purely by geometric constraints.
 Colloidal liquid crystals
Colloidal liquid crystals
Particles that are sufficiently non-spherical (either rod-like or plate-like) can form liquid crystalline phases, in particular a
nematic phase, on increasing concentration. For the first time, we have succeeded in obtaining such ordered suspensions starting from
a natural clay, sepiolite. A pronounced fractionation with respect to rod length is found, with the long rods accumulating in the nematic
phase.
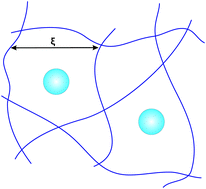 Attraction through repulsion
Attraction through repulsion
Presence of non-adsorbing polymer in solution induces a depletion attraction between suspended particles. This can lead to phase separation, aggregation
or gelation. This mechanism is relevant to practical formulations and addition of polymer allows a fine control of particle attractions. The mechanism also applies in the so-called protein limit, where the polymer chains are much larger than the suspended (nano-)particles. Addition of polymers is useful to help proteins crystallise. Recently we demonstrated phase separation and gelation induced by adding crosslinked polymer (microgel) particles.
-
A.I. Campbell, V.J. Anderson, J.S. van Duijneveldt, P. Bartlett,
Dynamical Arrest in Attractive Colloids: The Effect of Long-Range Repulsion,
Phys. Rev. Lett., 94: 208301, 2005.
-
Z. X. Zhang and J.S. van Duijneveldt,
Experimental Phase Diagram of a Model Colloid-Polymer Mixture in the Protein Limit,
Langmuir , 22: 63-66, 2006.
-
K.J. Mutch, J.S. van Duijneveldt, and J. Eastoe,
Colloid-protein mixtures in the protein limit,
Soft Matter 3: 155-167, 2007.
-
K.J. Mutch, J.S. van Duijneveldt, J. Eastoe, I. Grillo, and R.K. Heenan,
Small-Angle Neutron Scattering Study of Microemulsion-Polymer Mixtures in the Protein Limit,
Langmuir, 24: 3053-3060, 2008.
-
K.J. Mutch, J.S. van Duijneveldt, J. Eastoe, I. Grillo, and R.K. Heenan,
Testing the Scaling Behavior of Microemulsion-Polymer Mixtures,
Langmuir 25: 3944-3952, 2009.
-
K.J. Mutch, J.S. van Duijneveldt, J. Eastoe, I. Grillo, and R.K. Heenan,
Scaling the Structure Factors of Protein Limit Colloid-Polymer Mixtures,
Langmuir 26: 1630-1634, 2010.
-
J. Zhou, J.S. van Duijneveldt, and B. Vincent,
The Phase Behavior of Dispersions of Silica Particles in Mixtures of Polystyrene and Dimethylformamide,
Langmuir 26: 9397-9402, 2010.
-
K. Bayliss, J. S. van Duijneveldt, M. A. Faers and A. W. P. Vermeer,
Comparing colloidal phase separation induced by linear polymer and by microgel particles ,
Soft Matter 7, 10345-10352, 2011
Particle mixtures
Mixtures of repulsive colloidal particles of different shapes and sizes quite generally have a tendency to demix - with a variety of equilibrium and non-equilibrum structures resulting in practice.
-
N. Yasarawan and J.S. van Duijneveldt,
Arrested phase separation of colloidal rod�sphere mixtures,
Soft Matter, 6: 353-362, 2010.
-
J. Zhou, J.S. van Duijneveldt, and B. Vincent,
Two-stage phase separation in ternary colloid - polymer mixtures,
Phys. Chem. Chem. Phys. 13: 110-113, 2011.
-
J. Zhou, J.S. van Duijneveldt, and B. Vincent,
Phase separation in mixtures of two sizes of silica particles dispersed in
DMF on the addition of polystyrene,
Mol. Phys. 109: 1187-1194, 2011.
-
N. Doshi, G. Cinacchi, J. S. van Duijneveldt, T. Cosgrove, S. W. Prescott, I. Grillo,
J. Phipps and D. I. Gittins,
Structure of colloidal sphere�plate mixtures,
J. Phys.: Condens. Matter 23, 194109 (10pp), 2011
Unbinding books
Polymers and surfactants are used to prepare non-aqueous suspensions of clay platelets, in which the original clay
stacks are fully exfoliated into single sheets. High aspect ratio platelets can be obtained this way.
Small-angle X-ray scattering is a powerful technique to study these systems
at the nanometre length scale and is done in collaboration with
Prof.
Robert Richardson.
-
E.S.H. Leach, A. Hopkinson, K. Franklin and J.S. van Duijneveldt,
Non-aqueous suspensions of Laponite and montmorillonite,
Langmuir, 21: 3821-3830, 2005.
-
J. Connolly, J.S. van Duijneveldt, S. Klein, C. Pizzey, and R.M. Richardson,
Effect of surfactant and solvent properties on the stacking behaviour of
non-aqueous suspensions of organically modified clays,
Langmuir 22: 6531-6538, 2006.
-
Y. Cui and J.S. van Duijneveldt,
Adsorption of Polyetheramines on Montmorillonite at High pH,
Langmuir 26: 17210-17217, 2010.
-
Y. Cui, M. Threlfall, and J.S. van Duijneveldt,
Optimizing organoclay stabilized Pickering emulsions,
J. Colloid Interface Sci. 356: 665-671, 2011.
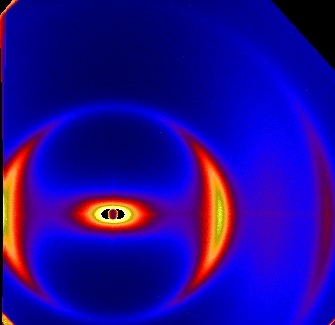 Liquid crystals as structured solvents
Liquid crystals as structured solvents
Liquid crystal suspensions consist of a (thermotropic) liquid crystal used as solvent for colloidal particles. In the nematic phase the presence
of the colloidal particles (spheres, rods, platelets) introduces defects in
the liquid crystalline host material which leads to novel particle interactions.
The X-ray scattering pattern on the left is from a clay suspension in 5CB. The nematic liquid crystal is
aligned in a magnetic field and gives rise to a diffuse scattering peak. The bright scattering peaks are due to the suspended clay platelets and
demonstrate that the platelets here are aligned perpendicular to the liquid crystal director.
-
C.
Pizzey, S. Klein, E. Leach, J.S. van Duijneveldt, and R.M. Richardson,
Suspensions of colloidal plates in a nematic liquid crystal: a small angle
X-ray scattering study, J. Phys.: Condens. Matter 16 (15): 2479-2495,
2004.
-
J.S. van Duijneveldt, S. Klein, E. Leach, C. Pizzey, R.M. Richardson,
Large scale structures in liquid crystal / clay colloids.
J. Phys.: Condens. Matter, 17: 2255-2267, 2005.
-
Z.X. Zhang and J.S. van Duijneveldt,
Effect of suspended clay particles on isotropic�nematic phase transition of liquid crystal,
Soft Matter 3: 596-604, 2007.
-
J. Connolly, J.S. van Duijneveldt, S. Klein, C. Pizzey and R.M. Richardson, Manipulation of modified clay particles in a nematic solvent by a magnetic field,
J. Phys.: Condens. Matter, 19: 156103 (17pp), 2007.
 Models
for (colloidal) liquid crystals
Models
for (colloidal) liquid crystals
Molecules or particles that are strongly non-spherical,
that is either rod-like or disc-like, tend to form liquid crystalline phases
that display a degree of order in between an isotropic liquid and a crystal.
We use Monte Carlo computer simulations to study how properties of the
molecules, such as their shape, flexibility and the presence of polar groups
influences the structure of the liquid crystalline phases and the phase
behaviour.
-
J.S.
van Duijneveldt and M. P. Allen. Free energy barriers for interlayer diffusion
in the smectic-A phase of hard spherocylinders. Mol. Phys.
90(2):243-250,
1997.
-
J.S.
van Duijneveldt and M.P. Allen, Computer simulation study of a flexible-rigid-flexible
model for liquid crystals, Mol. Phys. 92(5):855-870, 1997.
-
J.S.
van Duijneveldt, A. Gil-Villegas, G. Jackson, and M.P. Allen. Simulation
study of the phase behavior of a primitive model for thermotropic liquid
crystals: Rodlike molecules with terminal dipoles and flexible tails. J.
Chem. Phys. 112(20):9092-9104, 2000.
-
S.D.
Zhang, P.A. Reynolds and J.S. van Duijneveldt, Phase separation in
mixtures of colloidal platelets and non-adsorbing polymer: a scaled particle
treatment, Mol. Phys. 100(18):3041-3048, 2002.
-
S.D.
Zhang, P.A. Reynolds and J.S. van Duijneveldt, Phase behavior of mixtures
of colloidal platelets and nonadsorbing polymers, J. Chem. Phys.
117(21):9947-9958, 2002.
Methods
Projects in our group usually involve the preparation of model systems, such as colloidal spheres or platelets of well-defined size. A variety
of characterisation techniques are used, particularly scattering methods (light, X-rays, neutrons) and microscopy. Experimental work is
complemented by computer simulations. Much of the work is collaborative and funding comes from a range of sources including industry.
People
PhD students
- Laura Sosa Madrid - Clay nanocomposites
- Abraham Mauleon Amieva - Active matter
- Sian Fussell - Thermosensitive polymers
- Andrew Hambly - Clay-organic interactions
- Wendy Tang - Clay nanocomposites
- Wenfei Ji - Clay nanocomposite foams
Past group members
Postdoctoral fellows
- Giorgio Cinacchi - phase transitions in colloidal rods and platelets
- Aaron Olsen - dynamical arrest in colloidal model systems
- Andrew Campbell - colloidal crystallisation and confocal microscopy
- Valerie Anderson - colloidal crystallisation and confocal microscopy
- Shu-Dong Zhang - phase behaviour of mixtures of plate-like particles and polymers
- Gerrit Vliegenthart - gelation and phase separation of spherical particles
PhD students
- Nettraporn Doungsong - Hydrogels
- Ellen Pope - Nanofoams with auxetic behaviour
- William Ganley - clay suspensions
- Jessica Bonham - colloidal mixtures
- Claudia Ferreiro - colloidal rods and spheres
- Phillip Woolston - colloidal rods and platelets
- Emma Kastrisianaki-Guyton - nanomaterials
- Cathy Cooper - polymer adsorption
- Katie Bayliss - colloidal mixtures
- Yannan Cui - suspensions of clay platelets
- Juan Zhou - colloid - polymer mixtures
- Nisha Doshi - suspensions of clay platelets
- Yan Zhang - clay-polymer composites
- Nuttawisit Yasarawan - suspensions of rod-like clay particles
- Kevin Mutch - microemulsion - polymer mixtures
- Tim Kelly - use of polymers to control suspensions
- Zexin Zhang - colloidal spheres, rods and platelets
- Claire Pizzey - colloid - liquid crystal composites
- James Tait - colloidal particles suspended in liquid crystals
- Yves Hennequin - mixtures of sterically stabilised colloidal particles
- Paul Seymour - spherical particles with a thick stabilising polymer layer
- Edward Leach - non-aqueous clay suspensions
- James Weeks - aggregation of sterically stabilised colloids
Visitor
- Dr Wei Li, Capital Normal University, Beijing
Funding
We acknowledge financial support from the
University of Bristol, the Engineering and Physical Sciences Research Council
(EPSRC), the European Union, the Nuffield Foundation, the Department for
Education and Skills (DfES), the Mexican and Thai governments, Shell, Unilever, Hewlett-Packard, Bayer Cropscience, ICI / AkzoNobel, Imerys and Merck as well as beamtime allocations at Daresbury, ESRF, ILL and ISIS.
Curriculum vitae
Jeroen van Duijneveldt was appointed to a lectureship at the University of Bristol in 1997 and currently is reader in physical chemistry. He obtained
his Ph.D. in 1994 at the
Van
't Hoff Laboratory in Utrecht under supervision of Professor Henk Lekkerkerker
and Dr. Jan Dhont. Subsequently, he joined the group of
Professor Mike Allen at the Physics Department at the University of Bristol.
His publications are listed
here.
He is a fellow of the the Royal Society of Chemistry (RSC; CChem FRSC),
and member of the Royal Dutch Chemical Society, the Society of Chemical Industry and the Institute
of Physics (CPhys MInstP). He is
chair of the RSC Colloid and Interface Science Group and
member of the colloid and surface chemistry group of SCI (operating
jointly
)
and also past member and chairman of the
RSC Bristol & District Section Committee.
Return
to top
 Jeroen van Duijneveldt's research group
Jeroen van Duijneveldt's research group Soft condensed matter
Soft condensed matter Entropy driven gel formation and cluster phase
Entropy driven gel formation and cluster phase Colloidal liquid crystals
Colloidal liquid crystals Attraction through repulsion
Attraction through repulsion
 Liquid crystals as structured solvents
Liquid crystals as structured solvents Models
for (colloidal) liquid crystals
Models
for (colloidal) liquid crystals