![]()
Azelaic acid
The skin-whitening agent
that can be used to make Nylon
![]()
![]()
Molecule of the Month January 2026
Also available: HTML version.
![]()
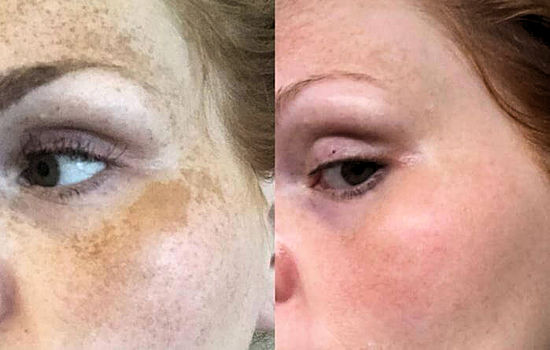
Melasma (brown skin patches) before and after treatment with azelaic acid.
Azelaic acidThe skin-whitening agent
|
 Melasma (brown skin patches) before and after treatment with azelaic acid. |
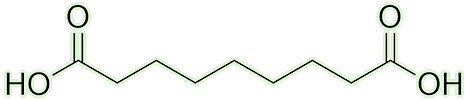
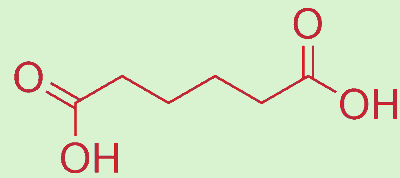
Anything to do with azaleas?No. The name azelaic acid actually derives from the fact that it was first produced by the oxidation of oleic acid (see MOTM for May 2006) with nitric acid. The "aze-" prefix comes from "azotic" (Greek for nitrogen) and the "-laic" part from the fact that it was also identified in rancid fats which contain short-chained smelly fatty acids, such as caprylic acid, butyric acid, propanoic acid, and which end with “-oic” or “-ic”. In fact, azelaic acid (AzA) is the dicarboxylic acid version of nonanoic acid (a.k.a. pelargonic acid). |
 Azaleas [Image: Missvain, CC BY 4.0, via Wikimedia Commons] |
 |
|
| Azelaic acid |
Well, industrially it is made by simply oxidising oleic acid with ozone. But in nature, AZA is made by a fungal yeast called Malassezia furfur. This yeast is naturally found on the skin surfaces of mammals and humans, and is responsible for a number of skin conditions such as seborrhoeic dermatitis and dandruff. But the yeast also degrades unsaturated fatty acids naturally present in the skin and converts them into C8-C12 dicarboxylic acids, including the C9 acid, AzA.

SEM image of the yeast Malassezia furfur.
[Image: CDC/Janice Carr public domain via Wikimedia Commons]
AzA is also found naturally in a number of plants, such as wheat, rye, and barley.
No, not them!
AzA acts like a signalling flare, which is produced when a plant is attacked or damaged by a pathogen, such as a bacteria, fungi, or insects. AzA then travels around the plant, warning other cells of the invasion, and getting them to switch into ‘war’ mode.
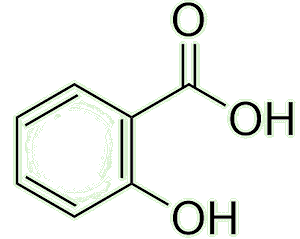
It causes the plant cells to synthesise and accumulate salicylic acid ...
 |
|
| Salicylic acid |
A precursor to aspirin, yes (see the MOTMs for Aspirin, Feb 1996 and methyl salicylate, December 2025) - which is used by the plants to fight off the attack.
Yes. Despite it causing skin conditions when synthesised by fungi on the skin, AzA is actually added to skin in the form of creams to treat skin conditions, such as acne. Acne is caused by a build up of dead skin cells and sebum (oil) which block the pores, and then attract bacteria, leading to rashes and pimples.
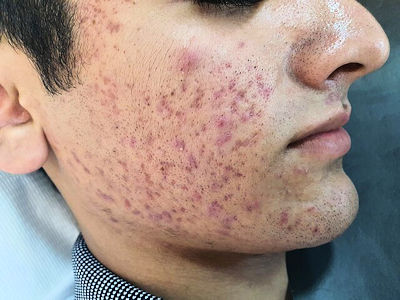 Acne [Image: Sedef94, CC BY-SA 4.0 via Wikimedia Commons] |
 Azelaic-Acid-based acne cream. [Image: Plinio Hurtado, CC BY-SA 4.0 via Wikimedia Commons] |
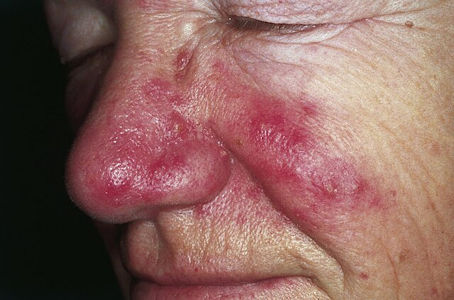 Rosacea seen over the cheeks and nose. [Image: PM. Sand, D. Sand, C. Thrandorf, V. Paech, P. Altmeyer, F. G. Bechara, CC BY 2.0 via Wikimedia Commons] |
AzA prevents this in a number of ways. First, it kills the bacteria that infect skin pores causing acne flare-ups. It also helps unblock the pores, preventing bacteria build up. Also, because AzA is an antioxidant, it can defend against the harmful effects of free radicals, preventing skin inflammation. Finally, AzA can help prevent the dilation of blood vessels in the skin surface, thereby minimizing any redness. It can also help with a condition called Rosacea.
Rosacea is a common skin condition in which the skin becomes red and sensitive. Once a person has Rosacea, there's no cure, only treatment. AzA can help with the red bumps and pimples that are caused by rosacea, and is usually administered in the form of a cream, gel, or foam – however care is needed when using the foam as this can be flammable! It’s also been reported that some foods and drinks might make the skin appear red or flushed while using AzA gel, so patients should avoid spicy foods, hot drinks (coffee and tea) and alcohol.
I heard it can be used as a skin-whitener, too?Yes, AzA can change the colour of skin, making it lighter and whiter. Up to 10% of skin cells produce a dark pigment known as melanin, and the more melanin in the skin the darker the skin colour (see MOTM for March 2016). In these cells, melanin is synthesised by the enzyme tyrosinase through a series of reactions involving the amino-acid, tyrosine. AzA and other C9-C11 dicarboxylic acids inhibit the activity of tyrosinase, stopping the production of melanin. Depigmentation and skin lightening products have been in use for many years in Asian countries where skin whiteness is a major cultural trend. Such whitening products have recently become highly valued by Western populations, too, due to excessive sun exposure. Azelaic acid is now commonly used for skin whitening, and is particularly useful in combatting melasma, dark, irregular, well-demarcated, patches on the skin, which can appear on the face or body, usually as a result of sun exposure. They can resemble large or numerous freckles, but in some cases can cover larger areas of the skin in unsightly brown regions. AzA, being a natural skin-bleaching agent, gives a paler skin, removing brown spots without significant side-effects and is much safer than some of the toxic alternatives, as you can see from the before-and-after picture at the top of this page. |
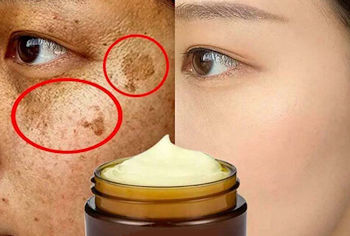 Whitening creams can remove melasma spots and make the skin paler. |
Yes, it is used to make Nylon!
Yes, well a type of Nylon (see MOTM for June 2010). The most common Nylon is Nylon 6,6, where both the monomers have 6 carbons. This is synthesised from a diamine (hexamethylene diamine - a 6-carbon chain with an amine group at each end) which reacts with a dicarboxylic acid (adipic acid – a 6-carbon chain with a carboxylic acid group at each end). The polymer formed, Nylon 6,6, has repeating units of 6 carbon atoms joined by amide bonds.
 |
 |
| Adipic acid | Hexamethylene diamene |
It is possible to replace either or both monomers with alternatives that have longer or shorter carbon chains, and this changes the properties of the Nylon produced.
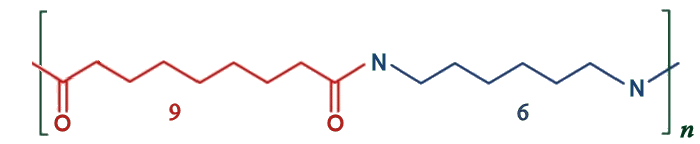
Nylon 6,9
AzA, with its 9-carbon chain, can be used together with hexamethylenediamene to make a Nylon with alternating 9 and 6 carbons, and so is called Nylon 6,9. Nylon-6,9 has some advantages over standard Nylon 6,6. It offers similar tensile yield and flexural strengths while having low water absorption, a low coefficient of friction, excellent electrical insulation properties, high resistance to chemicals, and a relatively high melting point. This high-performance engineering plastic is used in demanding industrial settings, especially for applications that involve exposure to high temperatures and where moisture might be problematic, such as wiring, cable insulation, and connectors.
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.30138973]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.30138973]