![]()
Lovastatin
(Mevacor)
The first statin in widespread use to lower cholesterol
![]()
Henry Goss-Custard
Eton College
![]()
Molecule of the Month April 2026
Also available: HTML version.
![]()

|
Lovastatin(Mevacor)The first statin in widespread use to lower cholesterol
Henry Goss-Custard
Molecule of the Month April 2026
|
 |
Lova-what? Something to do with Valentine's Day?No, but its connected with the heart, or more precisely heart attacks. |
 |
Tell me more.Over the course of human history, few health concerns have drawn as much attention and research as the effort to maintain a healthy circulatory system. And among the many factors influencing the function of this vital system, one molecule in particular has long been a focus: the waxy substance known as cholesterol (see MOTM for March 2014). Elevated levels of cholesterol are strongly linked to artery blockages and heart disease. Today, statins - of which lovastatin is one of the most well-known - are among the most effective treatments for lowering cholesterol and reducing the risk of cardiovascular disease. Yet their origins can be traced to an unexpected source: natural compounds that turned out to hold the key to blocking cholesterol production in the body. How was the first statin discovered?In the early 1970s, Japanese biochemist Akira Endo undertook a systematic investigation of fungi to identify natural compounds that might inhibit cholesterol synthesis. His hypothesis was that some species, in defending themselves against competing microbes, could produce substances able to interfere with an essential enzyme in cholesterol production. His team screened thousands of fungal strains before isolating a compound from Penicillium citrinum. This substance, later named mevastatin, was found to inhibit HMG-CoA reductase, which is the enzyme responsible for regulating cholesterol biosynthesis in the body. |
 Heart attacks are one of the most common causes of death in the world. [Image: Irfansevket2905, CC BY-SA 4.0 via Wikimedia Commons] |
 |
 |
| Akira Endo [Image: 文部科学省ホームページ, CC BY 4.0 via Wikimedia Commons |
Penicillium citrinum growing on an orange [Image: Stable Diffusion] |
Not quite. Mevastatin provided compelling evidence that fungal metabolites could interfere with cholesterol production, but its development was hampered by toxicity concerns. Even so, its discovery spurred other pharmaceutical groups, particularly those at Merck & Co., to pursue related compounds. Their efforts led to lovastatin, discovered in the fungus Aspergillus terreus and later refined to exhibit a safer clinical profile. Lovastatin would become the first statin approved for widespread use, and it fundamentally changed the way doctors treat heart disease.
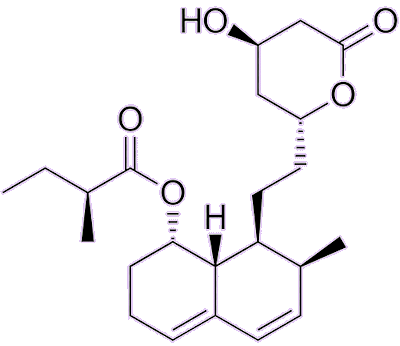 |
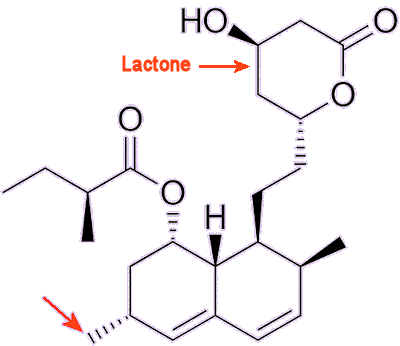 |
| Mevastatin [Image: Vaccinationist, via Wikimedia Commons |
Lovastatin The difference from mevastatin is the extra methyl group shown by the lower arrow. The lactone ring (a cyclic carboxylic ester) is shown by the upper arrow. |
So why do fungi produce cholesterol inhibitors in the first place?Fungi do not produce agents that target cholesterol per se; rather, they produce compounds that interfere with sterols more broadly, of which cholesterol is just one example. Sterols?Sterols are vital lipids that keep cell membranes stable and fluid across many organisms, including humans and fungi. The statins that fungi produce are actually targeted against ergosterol (see stucture, below), which is the fungal equivalent of cholesterol that many competing fungi rely on. In their natural environment, such as within soil, decaying organic matter, or even within other living organisms, fungi compete for limited resources. Blocking sterol biosynthesis in competing fungi compromises their membrane integrity and function, thereby impairing their capacity to survive or grow. |
 Aspergillus terreus [Image: Medmyco at English Wikipedia, CC0, via Wikimedia Commons] |
 |
|
| Ergosterol [Image: Mysid, via Wikimedia Commons] |
That's because the statin-producing fungi themselves tend to have versions of sterol-biosynthetic enzymes that are relatively insensitive to the inhibitors they secrete, and they tightly regulate intracellular exposure through compartmentalisation and active export.
Yes, statins have indeed been shown to exhibit antifungal activity, affecting a wide range of fungal species by interfering with ergosterol biosynthesis.
At this stage, the answer remains unclear, as all findings so far come only from limited laboratory studies. In clinical practice, however, it is doubtful that statin concentrations in the bloodstream would reach levels high enough to produce a meaningful antifungal effect.
Despite its somewhat negative popular reputation, cholesterol is an essential molecule in a range of biological processes. It forms a vital part of the structural framework of cell membranes, contributing to both their fluidity and stability. It also serves as a chemical precursor (a compound involved in any reaction that produces another compound) for steroid hormones (e.g. cortisol, oestrogen (MOTM for July 2019), testosterone (MOTM for August 2019)) and vitamin D (MOTM for November 2015), as well as being integral to bile acid formation in the liver.
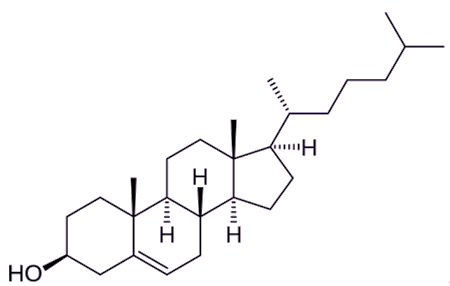 |
|
| Cholesterol [Image: BorisTM via Wikimedia Commons] |
Actually, the vast majority (around 80%) of the body's cholesterol is synthesised in the liver, while the remainder comes from dietary sources such as meat, dairy, and eggs. Under normal conditions, the body regulates internal cholesterol levels by modulating its own production to complement intake from the diet. However, factors like genetics, diet, and lifestyle frequently disrupt this balance, leading to elevated blood cholesterol (hypercholesterolaemia).

General structure of a plasma lipoprotein particle.
[Image: AntiSense via Wikimedia Commons]
Yes. Because cholesterol is insoluble in water it can only be transported through the bloodstream inside lipoprotein particles (see image above). These molecular complexes of lipids and proteins can transport any hydrophobic lipids, including triglycerides, fat-soluble vitamins, and cholesterol esters. They typically come in two main forms:
In addition to LDL and HDL, two further lipoprotein classes contribute to lipid transport in the circulation. Very-low-density lipoproteins (VLDL) are produced in the liver and transport both triglycerides and cholesterol to peripheral tissues. As triglycerides are progressively removed from the particle, VLDL is remodelled and eventually converted into LDL. In contrast, chylomicrons are formed in the intestine after a meal and carry dietary triglycerides and cholesterol into the bloodstream via the lymphatic system. Together, LDL, HDL, VLDL, and chylomicrons constitute the four principal lipoprotein classes responsible for distributing lipids throughout the body.
How does LDL block arteries?LDL tends to slip beneath inner lining of the arteries, often entering through areas that have existing damage caused by conditions, such as high blood pressure, smoking or diabetes. Once beneath the lining, LDL particles encounter free radicals and reactive oxygen species produced by surrounding cells - particularly endothelial cells (which make up the single cell layer that lines all blood vessels) and macrophages - leading to oxidation. Oxidised LDL is subsequently recognised as a foreign body by the immune system, triggering an inflammatory response that draws further macrophage immune cells into the artery wall. These macrophages engulf oxidised LDL and become foam cells (immune cells overloaded with LDL) that form visible fatty streaks along the artery wall. Smooth muscle cells subsequently migrate toward these fatty streaks to secrete fibrous proteins that stabilise the growing fatty deposits. This response is intended to restore structural integrity to the damaged vessel wall, but sustained lipid accumulation and ongoing inflammation gradually convert it into a fibrous atherosclerotic plaque that narrows and eventualy blocks the artery. What are the effects of exercise on cholesterol levels?Scientific evidence consistently supports exercise as an effective means of improving cholesterol balance. Two mechanisms account for this effect most clearly: increased activity of lipoprotein lipase (LPL) in skeletal muscle, and greater expression of the LDL receptor (LDLR) in the liver. When muscles contract, they draw on several fuels: glycogen, fatty acids released from adipose tissue, intramuscular triglycerides, and triglycerides carried in circulating lipoproteins such as VLDL and chylomicrons. Exercise stimulates signals that raise LPL activity in capillaries for many hours afterwards. LPL hydrolyses triglycerides from VLDL and chylomicrons, releasing fatty acids that muscle can oxidise. This enhanced activity accelerates clearance of triglyceride-rich lipoproteins from plasma, explaining the consistent fall in fasting triglyceride concentrations observed with regular training. Over time, this also alters LDL characteristics, with fewer small, dense LDL particles and a shift toward larger, more buoyant forms that are less atherogenic. |
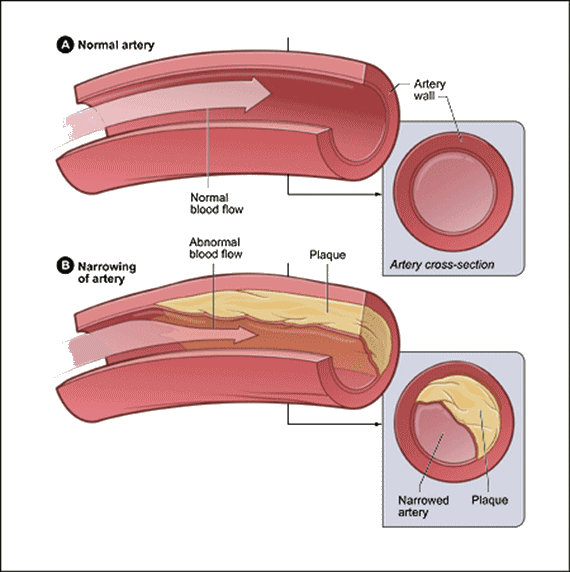 Comparison of a healthy artery (top) and one with plaque buildup (bottom). [Image: NHLBI, via Wikimedia Commons] |
Exercise simultaneously changes cholesterol handling in the liver. Regular training increases the use of cholesterol into synthesis of bile-acid , a pathway used both for fat digestion and as the main route for cholesterol excretion. As more cholesterol is channelled into this pathway, the hepatic (liver-based) cholesterol pool declines. The liver responds by increasing LDL receptor expression, which reduces circulating LDL cholesterol.
Clinically, having ‘high’ cholesterol usually means that concentrations of LDL or other atherogenic lipoproteins exceed guideline thresholds for a person’s level of cardiovascular risk. These thresholds vary depending on factors such as age, diabetes, blood pressure, smoking status, and prior cardiovascular disease.
By interfering with a key reaction that produces cholesterol in the body. Cholesterol is synthesised primarily in the liver through a multi-step pathway called the mevanolate pathway (see diagram, below). A sequence of enzymatic reactions converts acetyl-CoA (see MOTM for May 2007) into 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA). A key enzyme, HMG-CoA reductase, then catalyses the reduction of HMG-CoA to mevalonate, which is followed by further reactions to eventually yield cholesterol.
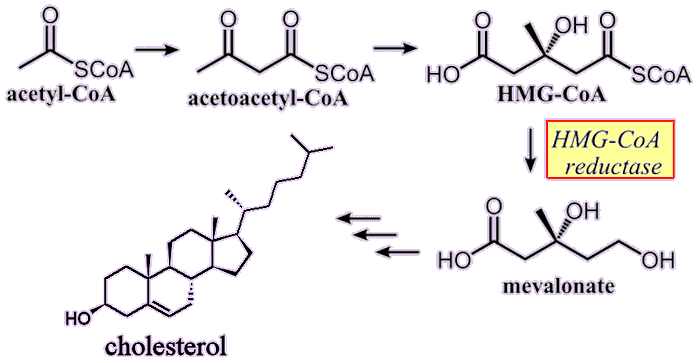
The mevanolate pathway which converts acetyl-CoA into cholesterol. Statins stop the enzyme HMG-CoA reductase from working, preventing the synthesis of cholesterol.
So what reaction do statins stop?Statins bind to the active site of HMG-CoA reductase, preventing the conversion of HMG-CoA to mevalonate and thereby reducing the liver’s capacity to synthesise new cholesterol. And all statins work the same way?Yes, but some statins, such as lovastatin, require activation before they can inhibit HMG-CoA reductase. They are administered in an inactive lactone form (a cyclic carboxylic ester), and undergo hydrolysis in the acidic environment of the stomach, opening the lactone ring and producing a pharmacologically active β-hydroxy acid (see structure, right). In its active β-hydroxy acid form, lovastatin binds to the active site of HMG-CoA reductase around 10,000 times more strongly than HMG-CoA, causing the enzyme to change shape and significantly impairing its function. As a result, cholesterol synthesis in the liver falls. Because cholesterol remains necessary for many crucial biochemicaly processes, such as membrane integrity and bile acid production, liver cells compensate for this decrease by trying to extract excess cholesterol from the blood supply. Crucially, they preferentially remove ‘bad’ LDL rather than ‘good’ HDL. The result is a fall in circulating LDL levels and, with it, a reduced risk of heart attack. |
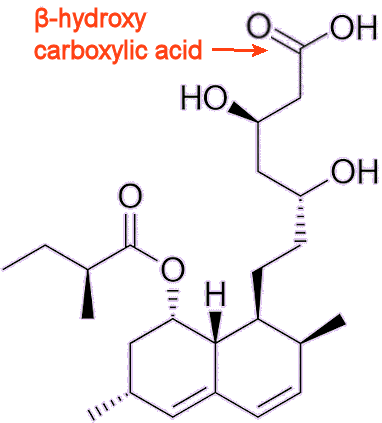 β-hydroxy carboxylic acid form of lovastatin |
 Deep-tank fermentation (Abbott Laboratories, UK, 1940s). |
And mass production?Once lovastatin’s promising cholesterol-lowering effects were established, the next challenge was to produce it on a large scale. The Merck & Co. researchers turned to fermentation-based methods, employing Aspergillus terreus grown in tightly regulated deep-tank fermenters, like those shown in the picture, left. The researchers optimised conditions such as temperature, nutrient composition, aeration, and pH to maximise the fungus’s output of lovastatin. Subsequent steps - filtration, extraction, and purification - were undertaken to ensure that the final product met both efficacy and safety standards. It was marketed by Merck & Co. under the brand name Mevacor. Following successful clinical trials, lovastatin gained approval from the U.S. Food and Drug Administration in 1987. Since then, additional statins have been developed - among them simvastatin, atorvastatin, and rosuvastatin - each with slightly differing pharmacokinetic properties and efficacy. Still, lovastatin continues to enjoy widespread use today. Can lovastatin besynthesised chemically ?Total synthesis of lovastatin is technically possible but commercially hopeless: it takes many tightly controlled, stereoselective steps with protecting-group manoeuvers, cryogenic conditions, and chromatography, resulting in low overall yields, long cycle times, high cost, and lots of waste. In contrast, fermentation using Aspergillus terreus builds the entire chiral scaffold in one bioprocess from cheap sugars, after which only minimal semi-synthetic tweaks are needed. On every process metric - yield, cost of goods, robustness, safety, and PMI - fermentation/semi-synthesis wins, and therefore total synthesis of bulk lovastatin is not considered desirable. This is yet another of the many cases where nature proves itself to be a much more adept chemist than mankind. |
Are statins safe?Research has shown that statins are generally safe and well-tolerated. Nonetheless, they can cause side-effects, the most commonly reported of which is muscle pain or weakness. The internet is full of sensationalist reports and videos of people claiming that statins are unsafe, causing a whole range of effects from muscle damage (rhabdomyolysis) to diabetes. But to counteract the internet misinformation and hype, a 2026 review in The Lancet showed that statins are far safer than once believed and do not actually cause most of the side-effects listed in patient leaflets (or on the internet!). The authors of the report say package leaflets should be updated to reflect this new evidence, and prevent people from being discouraged from using these life-saving drugs. The bottom line is that, in patients at moderate-to-high cardiovascular risk, the balance of evidence shows that the clinical benefits of statins substantially outweigh any potential harm. Has widespread adoption of statins caused a marked improvement in health outcomes?Indeed! Widespread statin use has contributed to better cardiovascular outcomes, especially in secondary prevention, where the benefit is strongest. Primary prevention refers to treating people who have not yet had a heart attack or stroke but are at elevated risk (for example, due to LDL level, age, diabetes, blood pressure, smoking, or family history) to reduce the chance of a first event. Secondary prevention is treating people with established cardiovascular disease or prior events (such as previous myocardial infarction, stroke, or known atherosclerotic disease) to reduce recurrence, complications, and mortality. |
 The 'dangers' of side-effects from statins are often over-hyped on the internet in videos like this one. |
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.31566856]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.31566856]