|

Methyl salicylate
The plant-signalling molecule
and minty painkiller found
in toothpaste and mouthwash

Simon Cotton
University of Birmingham

Molecule of the Month - December 2025
Also available: HTML version.

|
 |
Do I know it?
It’s got a minty smell and taste.
Where does it originate?
Notably in wintergreen (Gaultheria Procumbens); methyl salicylate is a key ingredient in oil of wintergreen, which is produced by the plant, though many other plants make it too.
Why do plants make it?
To talk to other plants.
|

Wintergreen (Gaultheria Procumbens)
[Image: John Delano of Hammond, Indiana, via Wikimedia Commons] |
 Pardon?
Pardon?
OK, here is an example. Scientists at the American National Centre for Atmospheric Research were studying a walnut grove, and found out that when the plants were stressed – due to drought and to widely fluctuating day-night temperatures – they emitted large amounts of methyl salicylate.
Why would they do that?
Methyl salicylate is quite a volatile molecule, which is emitted by one plant and absorbed by another, which might be several metres away. Once inside the other plant, it gets converted to the much less volatile salicylic acid, which acts as a signal for the plant to turn on its defence mechanisms and help them resist and recover from disease. You could call it a stress-related plant semiochemical (signalling chemical), if that is not too much of a mouthful.
Methyl salicylate has other uses in plants, too, this time to talk to friendly insects over longer distances, up to a hundred metres or so. For example, when spider mites attack lima beans, the plants release volatile organic compounds, including methyl salicylate, that attract predatory mites which eat the spider mites. The predatory mite Phytoseiulus persimilis is attracted by methyl salicylate to lima beans infested with the herbivorous mite Tetranychus urticae.
 |
 |
 |
Spider mite (Tetranychus urticae)
[Image: Ante Perkovic, CC BY-SA 3.0 via Wikimedia Commons] |
Lima bean plant (Phaseolus lunatus)
[Image: Filo gèn', CC BY-SA 4.0 via Wikimedia Commons] |
Phytoseiulus persimilis
[Image: Mick Talbot, CC BY 2.0 via Wikimedia Commons] |
Methyl salicylate has been found to repel aphids from bean and cereal crops. Plants also use methyl salicylate to warn each other of pathogenic infections – for example, tobacco plants inoculated with tobacco mosaic virus release it, to tell other plants to turn on their defences and activate disease resistance. A protein (tobacco SABP2) binds methyl salicylate and converts it into salicylic acid, whilst a gene for converting salicylic acid into methyl salicylate has also been identified.
Methyl salicylate is also used by plants for conventional attraction of pollinating insects. Electroantennogram responses of the White-lined Sphinx moth Hyles lineata to emissions from the plant fairy fans (Clarkia breweri) show a very strong response to methyl salicylate.
 |
 |
White-lined Sphinx moth (Hyles lineata)
[Image: Wildreturn, CC BY 2.0 via Wikimedia Commons] |
Fairy fans (Clarkia breweri)
[Image: Eric in SF, CC BY-SA 3.0 via Wikimedia Commons] |
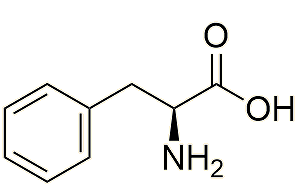
How do plants make methyl salicylate?
In several stages, each one catalysed by a different enzyme. One route starts from the amino acid phenylalanine, via trans-cinnamic acid. The salicylic acid is then esterified.
 |
 |
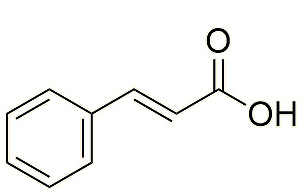 |
 |
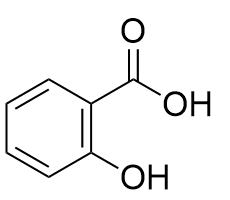 |
| Phenylalanine |
|
Trans-cinnamic acid |
|
Salicylic acid |
|
|
|
|
|
So why do we use it?
Apart from providing a minty taste to toothpaste and chewing gum, it is used in mouthwashes (like Listerine), and is an important ingredient in pain-relief embrocations, such as Ralgex and Deep Heat (UK), Tiger Balm, Icy Hot and Bengay (USA).

Can it be made in the laboratory?
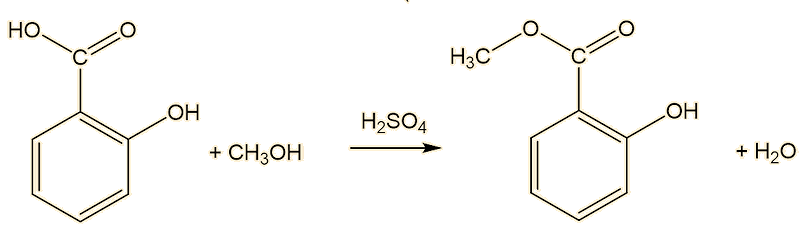
Indeed it can, by the classical method for making esters, the Fischer esterification reaction between methanol and salicylic acid, catalysed by a small amount of sulfuric acid.
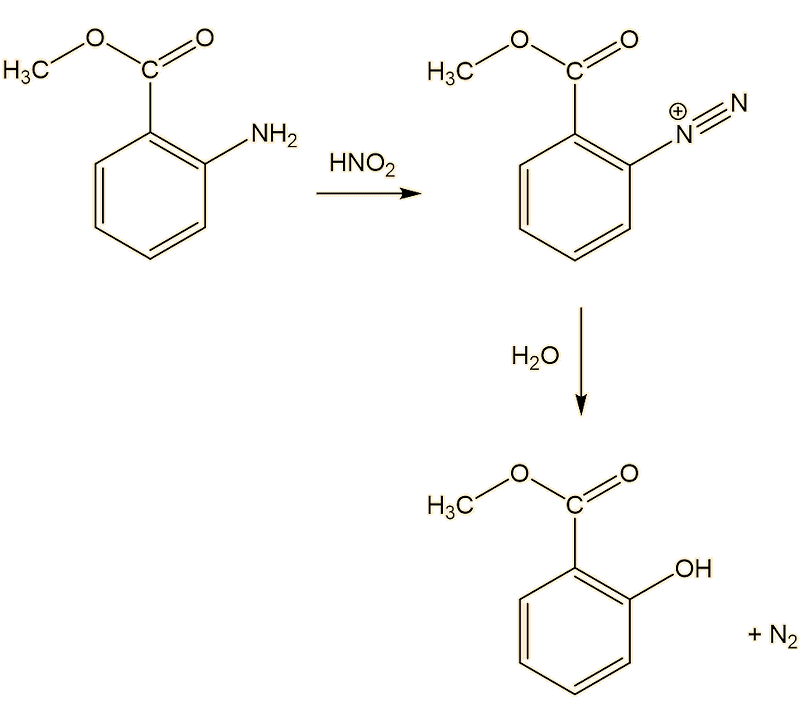
It can also be made by alternative routes. One starts from methyl anthranilate, carrying out a diazotisation and decomposing the diazonium ion in situ.
Another route starts from aspirin, and carries out a Fischer transesterification with methanol, using sulfuric acid catalyst.
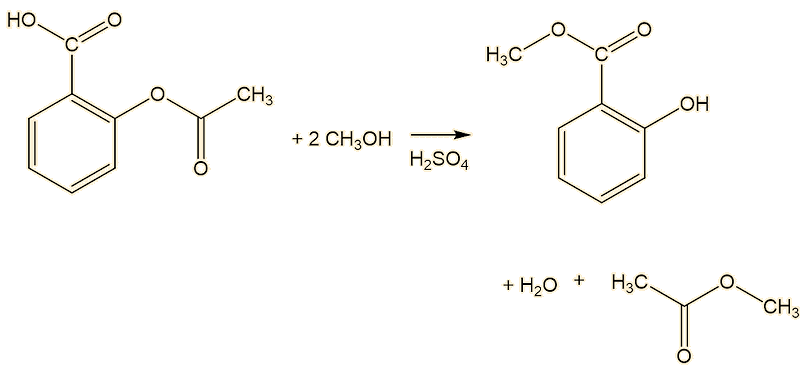
When tasted, it seems cooling. Why?
Methyl salicylate is one of a number of molecules which activate the TRPA1 (Transient receptor potential cation channel, subfamily A, member 1) channel, which gives rise to cold sensations. It also affects the TRPV1 channel, as do a number of noxious molecules like capsaicin (MOTM April 2001). Capsaicin is the chemical responsible for the ‘heat’ we associate with chilli peppers (the TRPV1 channel is popularly known as the ‘capsaicin receptor’). Methyl salicylate works because it first activates and then desensitises pain receptors. It has been found that a topical analgesic cream containing a mixture of methyl salicylate and L-menthol, which are cutaneous vasodilators, accelerates heat loss in exercise-induced hyperthermia, when it is applied to the skin during skin cooling.
Are there any problems with it?
Because it is found in over-the-counter medications, people assume that it must be harmless. Not so. In the body it is metabolised to salicylic acid. This is the active molecule in aspirin, acetyl salicylic acid (MOTM for February 1996).
 |
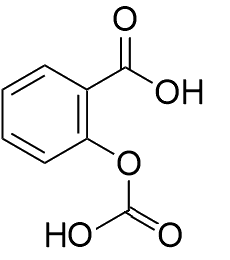 |
| Salicylic acid |
Acetylsalicylic acid
(Aspirin) |
|
|
|
Because it is absorbed through the skin, the methyl salicylate does not have to be swallowed to be toxic. It is reckoned that a ‘spoonful’ of methyl salicylate amounts to about 7 grams (equivalent to around 20 aspirin tablets), which could well be lethal to an adult, let alone a small child. Toxicity has also resulted from its application to the, er, genital region.
One fatality reported was that of an 80-year old man in the USA, who regularly had wintergreen oil rubbed into his legs by a carer. He mistakenly drank (but did not swallow) a mouthful of the oil, and subsequently died. The toxicity may have been exacerbated by the man suffering from end-stage renal disease.
Whilst known about for years, methyl salicylate poisoning became headline news in 2007, when Arielle Newman, a talented 17-year old athlete at Notre Dame Academy on Staten Island, New York, died from absorbing excessive amounts of methyl salicylate from a combination of a muscle-cream liberally applied to her legs, and from adhesive pads, which both contained the methyl salicylate. She apparently used this combination regularly and, together with the heating effect of exercise speeding up absorption, this appears to have caused the lethal dose. To quote the saying attributed to Paracelsus, ‘The dose makes the poison’. People using these medications must follow the manufacturer’s instructions, as with any other medication.
Who else uses methyl salicylate?
Male euglossine bees, often known as orchid bees, for one. These bees are unusual for several reasons – for a start they have iridescent bodies and long tongues. Found in the New World, they pollinate certain orchids and also collect small, fragrant, organic molecules – such as methyl salicylate, from sources like decaying wood and faeces, as well as the plants that they pollinate. They pick them up using specialised brushes on their forelegs, then rub the brushes against combs on their middle legs before pressing the combs onto grooves on the hind legs into a sponge-like cavity inside the hind tibia. It is thought that the bees then release these molecules at their display sites.
|

Denver Post article about Arielle Newman. |

Orchid bee
[Image: Eframgoldberg, CC BY-SA 3.0 via Wikimedia Commons]
 |
Is methyl salicylate anything to do with the 'Wint-O-Green Life Savers' Effect?
Ah, you mean the phenomenon that occurs when Wint-O-Green Life Savers sweets are crushed causing them to emit light? This is a popular game amongst children, who go somewhere dark, like a cupboard, and chew these sweets while keeping their mouths open. The triboluminescence that occurs as a result of the release of energy when the bonds in the sugar molecules are broken, causes sparks, and the children look like they have sparks inside their mouths. The Wint-O-Green Life Savers are particularly good for seeing this, because of the oil of wintergreen — methyl salicylate — flavouring used. Usually, most of the light emitted by fracturing sugar is in the ultraviolet, invisible to human eyes. But the methyl salicylate absorbs the ultraviolet light and re-emits the energy as blue-green light, which we see as flashes or sparks. |
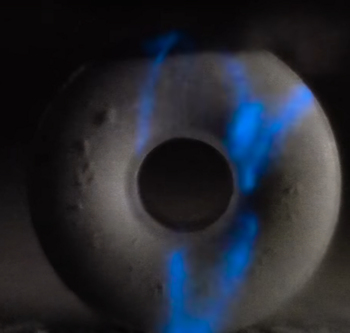
Still shot from a YouTube movie showing the triboluminescent flashes when a hammer crushes a Wint-O-Green Life Saver mint.
[Image: YouTube channel - Smarter Every Day]
|

Bibliography
Methyl salicylate
- Chapman and Hall Combined Chemical Dictionary code number: HHB30-H
Synthesis
- http://www.uccs.edu/Documents/chemistry/nsf/WintergreenExperiment.pdf
- https://www.apsu.edu/sites/apsu.edu/files/chemistry/SP11_1021_Synthesis_of_Aspirin_and_Oil_of_Wintergreen.pdf
- http://www.westfield.ma.edu/cmasi/organic_lab/organic1_lab/esterification_salicylic_acid.pdf
- M. Zanger and J. R. McKee, J. Chem. Educ., 1988, 65, 1106 (alternative route, from methyl anthranilate)
- E. Garcia Sega and J. Clarke, J. Chem. Educ., 2013, 90, 1658-1661 (alternative route, from aspirin)
Salicylic Acid Biosynthesis and Metabolism
- H.-I. Lee, J. Leon and I. Raskin, PNAS, 1995, 92, 4076-4079.
- J.-P. Métraux, Trends Plant Sci., 2002, 16, 356-362.
- D’ M. A. Dempsey, A. C. Vlot, M. C. Wildermuth and D. F. Klessig, The Arabidopsis Book, 2011, e156. doi: 10.1199/tab.0156
Methyl salicylate and pain relief
- M. Bandell, G. M. Story, S. W. Hwang, V. Viswanath, S. R. Eid, M. J. Petrus, T. J. Earley and A. Patapoutian, Neuron, 2004, 41, 849–857 (MeSal and TRPA1 channel)
- T. Ohta, T. Imagawa and S. Ito, Molecular Pharmacology, 2009, 75, 2307-317 (MeSal and TRPV1 channel)
- C. L. Goodnough, Y. Wu and E. R. Gross, Brit. J. of Anaesthesia, 2021, 127, E153-166 (BENGAY at work in rodents)
- I. de P. Ribeiro Argôlo, J.R. Parisi, J.R.T. da Silva and M.L. da Silva, J. Acupuncture and Meridian Stud, 2022, 15, 105-113 (MeSal and other TRPA agonists)
- G. Wang, T. Zhang, A. Wang and C. Hurr, Front. Physiol., 2022, 13, 945969 (methyl salicylate + L-menthol mixture accelerates heat loss during skin cooling)
- J. Guo, X. Hu, J. Wang, B. Yu, J. Li, J.Chen, X. Nie, Z. Zheng, S. Wang and Q. Qin, Front. Pharmacol., 2022, 13, 1015941 (methyl salicylate in compound linament for topical pain)
Methyl salicylate poisoning
- C. S. Stevenson, Am. J. Med. Sci., 1937, 193, 772–788 (case reports).
- R. A. MacCready, New Engl. J. Med., 1943, 228, 155-156 (case reports)
- M. M. Trill and M. L. Menten, Am. J. Dis. Child., 1945, 69, 37-43 (case reports).
- A. Danon, S. Ben-Shimon and Z. Ben-Zvi, Eur. J. Clin. Pharmacol., 1986, 31, 49-52 (effect of exercise and heat exposure on percutaneous absorption of methyl salicylate)
- J. R. Brubacher and R. S. Hoffman, J. Toxicol. Clin. Toxicol., 1996, 34, 431-436 (salicylism from topical salicylates: review of the literature)
- T. Y. K. Chan, Hum. Exp. Toxicol., 1996, 15, 747-750 (dangers from topical preparations containing methyl salicylate)
- T. Y. K. Chan, Postgrad Med J., 1996, 72, 109-112 (risk of severe salicylate poisoning from topical medicaments or aspirin)
- L. Mason, R. A. Moore, J. E. Edwards, H. J. McQuay, S. Derry and P. J. Wiffen, BMJ, 2004, 328, 995-997 (review of efficacy of topical rubefacients containing salicylates for pain treatment)
- R. L. Chin, K. R. Olson and D. Dempsey, Calif. J. Emerg. Med., 2007, 8, 23-25 (death of 80 year old American man)
- W. L. Yip, H. W. Ng, Y. C. Chan, M. L. Tse and F. L. Lauo, Hong Kong J. Emerg. Med., 2010, 17, 54-57 (blood levels of methyl salicylate from skin absorption)
- T. M. Thompson, T. Toerne and T. B. Erickson, West. J. Emerg. Med., 2016, 17, 181-183 (toxicity from genital exposure)
- https://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+1935
- http://www.washingtonpost.com/wp-dyn/content/article/2007/06/15/AR2007061500676.html
- http://www.nbcnews.com/id/19208195/ns/health-fitness/t/sports-cream-warnings-urged-after-teens-death/#.V1_aAvkrLcs
Plant signalling
- R. A. Raguso, D. M. Light and E. Pichersky, J. Chem. Ecol., 1996, 22, 925-934 (electroantennogram responses of the moth Hyles lineata to volatile compounds including methyl salicylate)
- V. Shulaev, P. Silverman and I. Raskin, Nature, 1997, 385, 717-721 (tobacco plants inoculated with tobacco mosaic virus emit methyl salicylate)
- G-i. Arimura, R. Ozawa, T. Shimoda, T. Nishioka, W. Boland and J. Takabayashi, Nature, 2000, 406, 512–515 (spider-mite predators cause lima-bean leaves to emit methyl salicylate)
- F. Chen, J.C. D’Auria, D. Tholl, J. R. Ross, J.Gershenson, J. P. Noel and E. Pichersky, Plant J., 2003, 36, 577-588 (gene for methyl salicylate biosynthesis)
- J. G. De Boer and M. Dicke, J. Chem. Ecol., 2004, 30, 255-271 (predatory mites use MeSal in searching for prey)
- F. Forouhar, et al., PNAS, 2005, 102, 1773–1778 (SA-binding protein 2 (SABP2) from tobacco)
- T. Karl, A. Guenther, A. Turnipseed, E. G. Patton and K. Jardine, Biogeosciences, 2008, 5, 1287–1294. (walnut trees under stress produce methyl salicylate)
- M. Šimpraga, J. Takabayashi and J. K. Holopainen, J. Integ. Plant Biol., 2016, 58, 343–349 (review)
- D’M. A. Dempsey and D. F. Klessig, BMC Biology, 2017, 15, 23 (review on how salicylic acid combats disease in plants)
Orchid bees
- D. L. Pearson and R. L. Dressler, J. Trop. Ecol., 1985, 1, 37-54 (Peruvian orchid bees’ attraction to chemical baits)
- W. S. Armbruster, Biotropica, 1993, 25, 122-128 (euglossine bees’ attraction).
- F. P. Schiestl and D. W. Roubik, J. Chem. Ecol., 2003, 29, 253-257 (euglossine bees).
- S. A. Cameron, Annu. Rev. Entomol. 2004, 49, 377–404 (euglossine bees).
- C. Skov and J. Wiley, Fla. Entomol., 2005, 88, 225–227 (euglossine bees in Florida)
- H. Hentrich, R. Kaiser and G. Gottsberger, Ecotropica, 2007, 13, 149–155 (floral scent collection by orchid bees)
- M. G. Ferreira, O.C. de Pinho, J.B.P. Balestieri and O. Faccenda, Neotrop. Entomol., 2011, 40, 639-646.


 Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.26840614]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.26840614]

![]()
![]()
![]()
![]()


 Pardon?
Pardon?