
![]()
NITRIC ACID
The starting point for explosives and fertilisers"
![]()
Paul May
University of Bristol, Bristol, UK
![]()
Molecule of the Month November 2007
Also available: HTML version.
![]()
 |
NITRIC ACIDThe starting point for explosives and fertilisers"
Paul May
Molecule of the Month November 2007
|
 |
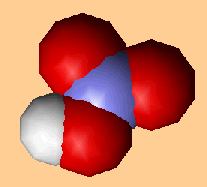
Nitric acid (HNO3) is an extremely important chemical used in the manufacture of fertilisers and explosives. It is made from ammonia by the Ostwald Process which was developed in 1902 by the German chemist Wilhelm Ostwald (below, left), who got the Nobel prize for Chemistry in 1909. This process reacts together O2 and ammonia NH3 at 850�C and 5 atmospheres pressure, with the help of platinum and rhodium catalysts, to make NO gas. This is then oxidised to NO2, which is then dissolved in water to make HNO3.
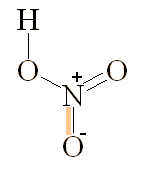 |
|
| Nitric acid |
 The Ostwald process was discovered just in time for the First World War, and it contributed greatly to the extended length of that war. This is because previously Germany had no nitrate deposits of its own from which to make the nitric acid that was essential for the production of the explosives used in artillery shells, such as TNT and nitroglycerine. In fact, most of the nitrates were only available from guano, which is the droppings of fish-eating sea birds, and is found in large quantities on the islands off the coast of Peru. When hostilities began, the shipping routes to Germany across the Atlantic were blocked, and Germany needed a new method to make nitric acid.
The Ostwald process was discovered just in time for the First World War, and it contributed greatly to the extended length of that war. This is because previously Germany had no nitrate deposits of its own from which to make the nitric acid that was essential for the production of the explosives used in artillery shells, such as TNT and nitroglycerine. In fact, most of the nitrates were only available from guano, which is the droppings of fish-eating sea birds, and is found in large quantities on the islands off the coast of Peru. When hostilities began, the shipping routes to Germany across the Atlantic were blocked, and Germany needed a new method to make nitric acid.
 Coincidentally, again just before WW1 started, another German chemist, Fritz Haber, had found a method to turn inert nitrogen gas from air into ammonia. By 1913, the German chemical giant BASF (Badische Aniline und Soda Fabrik) had a plant operating in Ludwigshaven-Oppau, Germany making ammonia at the rate of 30 metric tons per day. Without question, the Haber-Bosch process for turning N2 into NH3, combined with the Ostwald process for turning NH3 into nitric acid, permitted Germany to continue making explosives, and extended the war for many years. Some people say that without these two chemical processes, WW1 might never have happened at all.
Coincidentally, again just before WW1 started, another German chemist, Fritz Haber, had found a method to turn inert nitrogen gas from air into ammonia. By 1913, the German chemical giant BASF (Badische Aniline und Soda Fabrik) had a plant operating in Ludwigshaven-Oppau, Germany making ammonia at the rate of 30 metric tons per day. Without question, the Haber-Bosch process for turning N2 into NH3, combined with the Ostwald process for turning NH3 into nitric acid, permitted Germany to continue making explosives, and extended the war for many years. Some people say that without these two chemical processes, WW1 might never have happened at all.
 At room temperature, 100% pure anhydrous nitric acid is a liquid, but becomes a colourless white solid below -41°C and boils at 83°C. What we call 'concentrated nitric acid' is actually a solution of 68% by weight HNO3 in water (a 16M solution), and is often pale yellow as a result of photochemical decomposition which releases NO2. Dissolving even more NO2 into the pure material produces red 'fuming' nitric acid, which is an extremely powerful acid and oxidising agent using in the semiconductor industry for cleaning silicon wafers. The acid aqua regia (approx 3 vols HCl to 1 vol HNO3) contains free Cl2 and nitrosyl chloride (NOCl). This powerful acid attacks even the inert metals gold and platinum owing to the ability of Cl- to stabilise the complexes AuCl4- and PtCl62-.
At room temperature, 100% pure anhydrous nitric acid is a liquid, but becomes a colourless white solid below -41°C and boils at 83°C. What we call 'concentrated nitric acid' is actually a solution of 68% by weight HNO3 in water (a 16M solution), and is often pale yellow as a result of photochemical decomposition which releases NO2. Dissolving even more NO2 into the pure material produces red 'fuming' nitric acid, which is an extremely powerful acid and oxidising agent using in the semiconductor industry for cleaning silicon wafers. The acid aqua regia (approx 3 vols HCl to 1 vol HNO3) contains free Cl2 and nitrosyl chloride (NOCl). This powerful acid attacks even the inert metals gold and platinum owing to the ability of Cl- to stabilise the complexes AuCl4- and PtCl62-.
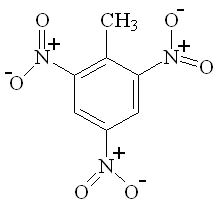
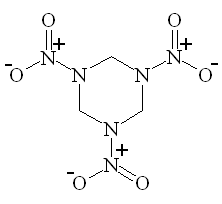
As noted above, one important use for nitric acid is the manufacture of various organic nitro compounds, especially explosives, such as trinitrotoluene (TNT), nitrocellulose, nitroglycerin, and RDX and PETN (the last two being components of Semtex). For example, nitroglycerine (the explosive component of dynamite) is made by adding nitric and sulphuric acids to glycerol under very carefully controlled conditions. The nitrating agent is the NO2+ ion, which can attach to organic chains and rings to give a molecule with high potential energy. These molecules are often strained, and since they contain an in-built supply of oxygen, and N, they readily decompose (explosively) to give CO2 and N2, releasing a lot of heat very rapidly.
 |
 |
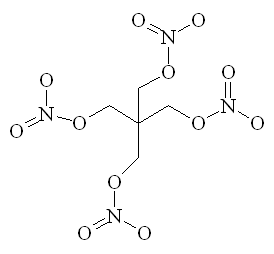 |
| TNT | RDX | PETN |
| R |
 Nitrates
NitratesThe biggest (80%) use of nitric acid, however, is in making ammonium nitrate, NH4NO3. This is an ingredient in many gunpowder recipes, and is an important explosive in its own right, but is mainly used as an agricultural fertiliser. It is also the parent of many other nitrates (compounds containing the NO3- ion), which are very rarely found in natural rocks since their high solubility in water means any deposits simply get washed away. Although such nitrate fertilisers have provided great benefits to mankind in increasing the food yield from otherwise poor soils, they have recently received a good deal of bad press. This is, again, due to their high solubility, which means the excess nitrates are readily washed into streams. There, they continue their job of promoting the growth of plantlife, particularly algae which reproduce unchecked to form smothering masses. The respiration of the algal masses use up the oxygen in the water, suffocating all other plant and animal life (see photo, right).
 Nitrites, (compounds containing the NO2- ion), on the other hand, are made from nitrous acid (HNO2), which is a very weak acid compared to its close relative, nitric acid. For example, sodium nitrite, NaNO2 (E250) and potassium nitrite, KNO2 (E249) are used as a preservative in several processed meats, such as bacon, ham, sausage, corned beef, beef jerky, frankfurters, and some fish products. The nitrite ions (NO2-) in these meats inhibit the growth of a bacterium (Clostridium botulinum) which causes the fatal food poisoning known as botulism. They are also used because they fix the bright-red colour of fresh meat (in the form of a type of diazo-dye), which would otherwise quickly fade to an unpalatable brown. This means that meat on the supermarket shelves, which looks fresh and new, may actually be several days old and just 'dyed' red using the nitrite!
Nitrites, (compounds containing the NO2- ion), on the other hand, are made from nitrous acid (HNO2), which is a very weak acid compared to its close relative, nitric acid. For example, sodium nitrite, NaNO2 (E250) and potassium nitrite, KNO2 (E249) are used as a preservative in several processed meats, such as bacon, ham, sausage, corned beef, beef jerky, frankfurters, and some fish products. The nitrite ions (NO2-) in these meats inhibit the growth of a bacterium (Clostridium botulinum) which causes the fatal food poisoning known as botulism. They are also used because they fix the bright-red colour of fresh meat (in the form of a type of diazo-dye), which would otherwise quickly fade to an unpalatable brown. This means that meat on the supermarket shelves, which looks fresh and new, may actually be several days old and just 'dyed' red using the nitrite!
However, there is controversy at the moment as to the toxicity of these nitrite additives, since it is known that when nitrite-cured meats are fried or boiled they can produce nitrosamines (compounds containing -NH-NO- groups) which are among the most powerful cancer-causing agents known. The counter argument is that removal of the nitrite preservatives would result in epidemics of food poisoning. The result of this controversy has been a reduction in the allowed concentration of nitrites in meat products, however it has still to be proven that nitrites cause cancer in animals.
![]()
![]()