


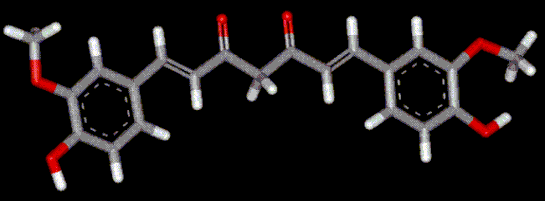
 Curcumin provides a water soluble orange-yellow colour.
It is a natural extract obtained by solvent extraction from the dried rhizomes of turmeric (used in Indian cuisine as a flavouring agent). Curcumin may be used to compensate for fading of natural colouring in pre-packed foods. Recognised as an anticarcinogenic agent during laboratory tests.
Curcumin provides a water soluble orange-yellow colour.
It is a natural extract obtained by solvent extraction from the dried rhizomes of turmeric (used in Indian cuisine as a flavouring agent). Curcumin may be used to compensate for fading of natural colouring in pre-packed foods. Recognised as an anticarcinogenic agent during laboratory tests.| Other names: | 1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione. |
| Molecular formula: | [HOC6H3(OCH3)CH:CHCO]2CH2 |
| CAS No: | 458-37-7 |
| Physical appearance: | orange crystalline powder |
| Melting point: | 180 C |
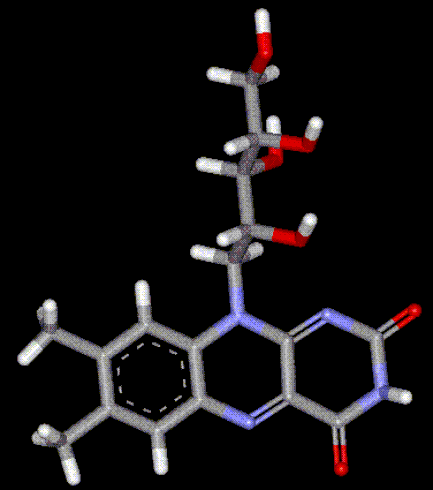 Riboflavin is an essential dietary requirement, as it aids in the metabolism of fats, carbohydrates and proteins. It is also needed for other functions including red blood cell formation, respiration, antibody production and general well-being. Activation of vitamin B6 and folic acid require riboflavin. Riboflavin has been found to aid in the treatment of eye disorders, e.g. cataracts. It is found naturally in liver, kidneys, eggs, milk but is destroyed upon exposure to light. It is manufactured industrially using yeast or other fermenting organisms, used as a yellow colouring and as vitamin fortification, but is difficult to incorporate into most foods due to poor solubility.
Riboflavin is an essential dietary requirement, as it aids in the metabolism of fats, carbohydrates and proteins. It is also needed for other functions including red blood cell formation, respiration, antibody production and general well-being. Activation of vitamin B6 and folic acid require riboflavin. Riboflavin has been found to aid in the treatment of eye disorders, e.g. cataracts. It is found naturally in liver, kidneys, eggs, milk but is destroyed upon exposure to light. It is manufactured industrially using yeast or other fermenting organisms, used as a yellow colouring and as vitamin fortification, but is difficult to incorporate into most foods due to poor solubility.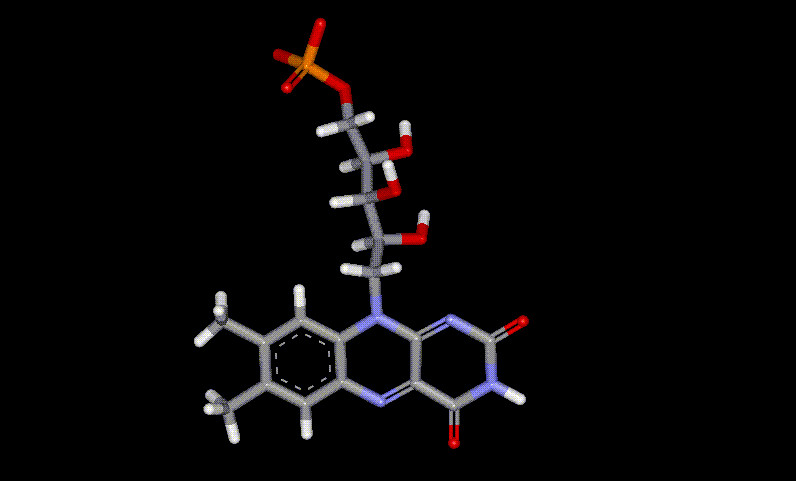 Riboflavin-5'-phosphate is chemically prepared from Riboflavin (converted to free Riboflavin upon ingestion), and is used to provide vitamin B2 enrichment, also a yellow colour. It may be used in preference to Riboflavin due to better solubility, however it is more expensive.
Riboflavin-5'-phosphate is chemically prepared from Riboflavin (converted to free Riboflavin upon ingestion), and is used to provide vitamin B2 enrichment, also a yellow colour. It may be used in preference to Riboflavin due to better solubility, however it is more expensive.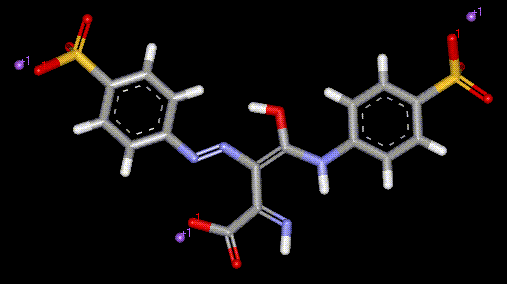 Tartrazine is a yellow synthetic azo dye, and is responsible for a large number of allergic reactions including migraine, blurred vision, itching, rhinitis and skin blotching. Asthmatics and aspirin intolerant sufferers appear to be particularly at risk. Tests have shown that in large doses, tartrazine will raise the plasma histamine levels of healthy adults. There is a suspected link between hyperactivity in children and tartrazine. Other commercial uses include as a 0.01% solution in EtOH for photometric determination of Pd, a dye for wool and silk.
Tartrazine is a yellow synthetic azo dye, and is responsible for a large number of allergic reactions including migraine, blurred vision, itching, rhinitis and skin blotching. Asthmatics and aspirin intolerant sufferers appear to be particularly at risk. Tests have shown that in large doses, tartrazine will raise the plasma histamine levels of healthy adults. There is a suspected link between hyperactivity in children and tartrazine. Other commercial uses include as a 0.01% solution in EtOH for photometric determination of Pd, a dye for wool and silk.| Other names: | 4,5-Dihydro-5-oxo-1-(4-sulfophenyl)-4-[(4-sulfophenyl)azo]-1H-pyrazole- 3-carboxylic acid 5-Hydroxy-1-(4-sulfophenyl)-4-[(4-sulfophenyl)azo]pyrazole-3-carboxylic acid |
| Physical appearance: | Crystalline. |
 dione, and the product is essentially sodium salts of a mixture of disulfonates, monosulfonates and trisulfonates of the dione. The calcium and potassium salts may also be permitted. Quinoline Yellow is used to give a yellow to greenish yellow colour. May induce an allergic reaction.
dione, and the product is essentially sodium salts of a mixture of disulfonates, monosulfonates and trisulfonates of the dione. The calcium and potassium salts may also be permitted. Quinoline Yellow is used to give a yellow to greenish yellow colour. May induce an allergic reaction.| Chemical formula: | C18H9N Na2O8S2 (principal component) |
| Physical appearance: | Yellow powder. |
 Yellow 2G is a synthetic 'coal tar' dye and also an azo dye, used to give a yellow colour to food, although the EEC is proposing a total ban as there are links between Yellow 2G and allergic reactions, particularly amongst those suffering from asthma or aspirin intolerance. It is also used commercially in printing inks.
Yellow 2G is a synthetic 'coal tar' dye and also an azo dye, used to give a yellow colour to food, although the EEC is proposing a total ban as there are links between Yellow 2G and allergic reactions, particularly amongst those suffering from asthma or aspirin intolerance. It is also used commercially in printing inks.| Chemical formula: | HOC6H3(CH3)N=NC6H4NHC(O)CH3 |
| Other names: | Disodium 2-hydroxy-1-(4-sulfonatophenylazo)napthalene-6-sulfonate |
| Chemical formula: | C16H10N2Na2O7S2 |
 Cochineal is a natural red colour obtained by crushing of the female Dactilopius coccus, a cactus-dwelling insect indigenous to Central America. The dye is expensive due to the sheer quantity of shells required to produce a small amount. Alcoholic drinks may contain the water soluble form (ammonium carmine), but the insoluble calcium carmine is found in a many more products. Other commercial uses include as an antineoplastic agent, as a 0.025% solution in concentrated H2SO4 for photometric and fluorimetric determination of B, as an indicator and diagnostic agent (gives colour reactions with Mg, Ge, Pb, Zr, Th, Mo, U). E120 has been linked to the cause of allergic reactions.
Cochineal is a natural red colour obtained by crushing of the female Dactilopius coccus, a cactus-dwelling insect indigenous to Central America. The dye is expensive due to the sheer quantity of shells required to produce a small amount. Alcoholic drinks may contain the water soluble form (ammonium carmine), but the insoluble calcium carmine is found in a many more products. Other commercial uses include as an antineoplastic agent, as a 0.025% solution in concentrated H2SO4 for photometric and fluorimetric determination of B, as an indicator and diagnostic agent (gives colour reactions with Mg, Ge, Pb, Zr, Th, Mo, U). E120 has been linked to the cause of allergic reactions.| Other names: | C. I. Natural Red 4 |
| Chemical formula: | C22H20O13 |
| CAS NO.: | 1260-17-9 |
| Physical appearance: | bright red powder |
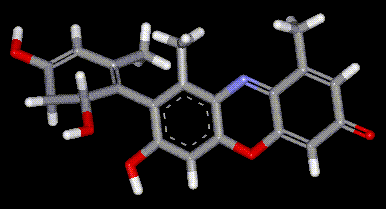
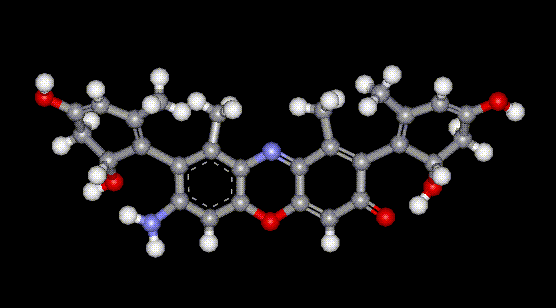
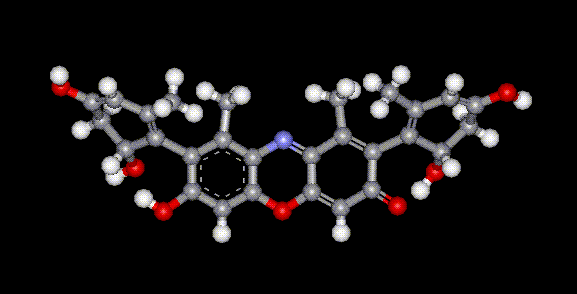
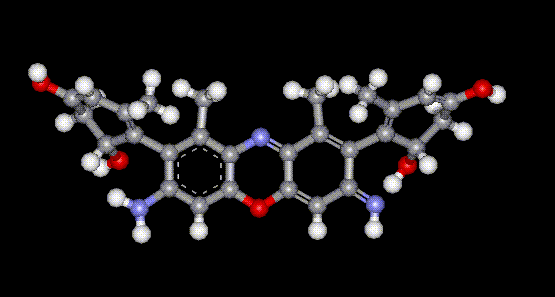
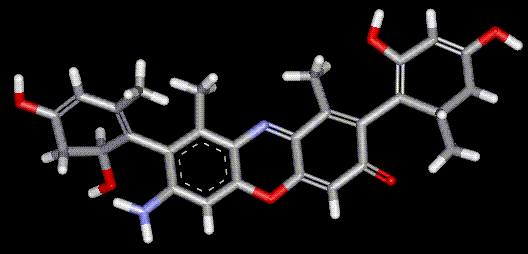
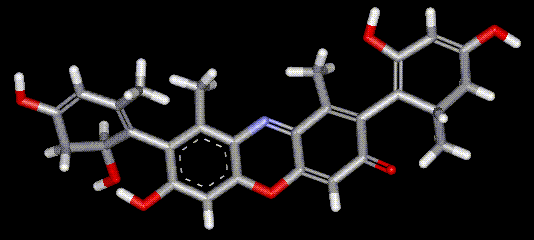
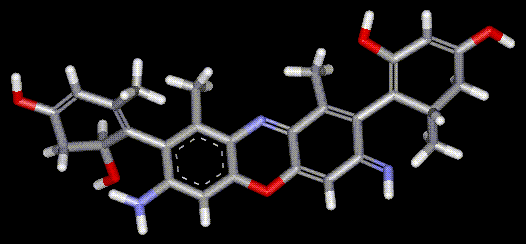 Orcein and orchil are colourings derived from archil, the lichen Rocella tinctoria. Orcinol is derived from the lichen and then converted to orcein (a reddish-brown dye, also used as a microscopical stain) by the action of aqueous ammonia and air. Orchil is a purple-blue dye. Orcein is a mixture of compounds with a phenoxazone structure, composed of hydroxy-orceins, amino-orceins and amino-orceinimines (see molecular diagrams). Hans Musso (1925-1988) published many papers about orcein.
Orcein and orchil are colourings derived from archil, the lichen Rocella tinctoria. Orcinol is derived from the lichen and then converted to orcein (a reddish-brown dye, also used as a microscopical stain) by the action of aqueous ammonia and air. Orchil is a purple-blue dye. Orcein is a mixture of compounds with a phenoxazone structure, composed of hydroxy-orceins, amino-orceins and amino-orceinimines (see molecular diagrams). Hans Musso (1925-1988) published many papers about orcein.| Other names: | C.I. natural red 28 |
| Molecular formula: | C28H24N2O7 |
| CAS No: | 1400-62-0 |
| Physical appearance: | dark brown crystals |
| Other information: | Harmful if swallowed, inhaled or absorbed through the skin. |
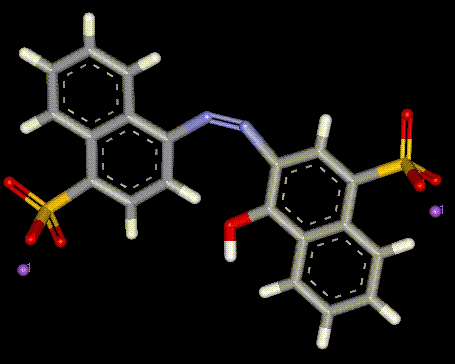 Carmoisine is a synthetic azo dye used to give a red colour to foods, especially those which require heat treatment. There may be adverse reactions to this dye from people with asthma or aspirin intolerance, including nettle rash and water retention. It is also used in photometric determination of Mg, Pd, Cu, Sn, Cr.
Carmoisine is a synthetic azo dye used to give a red colour to foods, especially those which require heat treatment. There may be adverse reactions to this dye from people with asthma or aspirin intolerance, including nettle rash and water retention. It is also used in photometric determination of Mg, Pd, Cu, Sn, Cr.| Chemical formula: | C20 H14 N2 O7 S2 . 2 Na (Derived) |
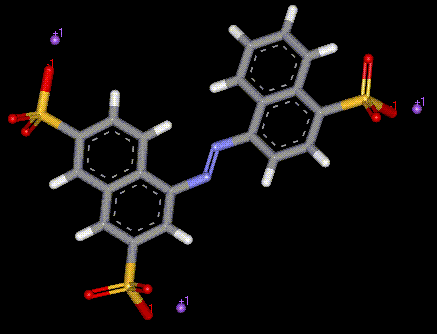 Amaranth is a purple-red synthetic coal tar or azo dye, which in addition to colouring in food, is used in dyeing and colour photography. As amaranth is an azo dye it is recommended that people who suffer from asthma or aspirin intolerance avoid it. Amaranth may cause a skin rash.
Amaranth is a purple-red synthetic coal tar or azo dye, which in addition to colouring in food, is used in dyeing and colour photography. As amaranth is an azo dye it is recommended that people who suffer from asthma or aspirin intolerance avoid it. Amaranth may cause a skin rash.
| Chemical formula: | C20 H14 N2 O10 S3 . 3 Na (Derived) |
| Appearance: | dark red-brown powder. |
Other names:| Trisodium 2-hydroxy-1-(4-sulfonato-1-naphthylazo)
naphthalene-3,6-disulfonate. |
|
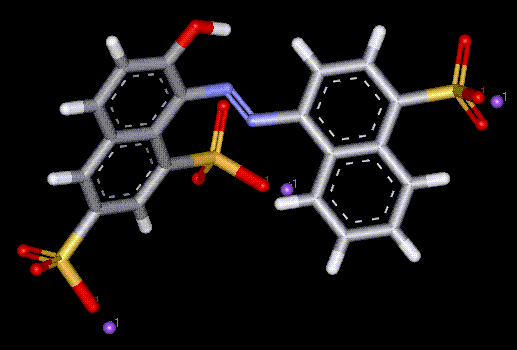 Ponceau 4R is a red synthetic coal tar and azo dye, which appears to cause an allergic reaction in sensitive people, particularly asthma sufferers and those intolerant to aspirin.
Ponceau 4R is a red synthetic coal tar and azo dye, which appears to cause an allergic reaction in sensitive people, particularly asthma sufferers and those intolerant to aspirin.
| Chemical formula: | C20H14N2O10S3 |
| Physical appearance: | bright red powder. |
Other names:| 7-Hydroxy-8-[(4-sulfo-1-naphthalenyl)azo]-1,3-naphthalenedisulfonic acid. |
|
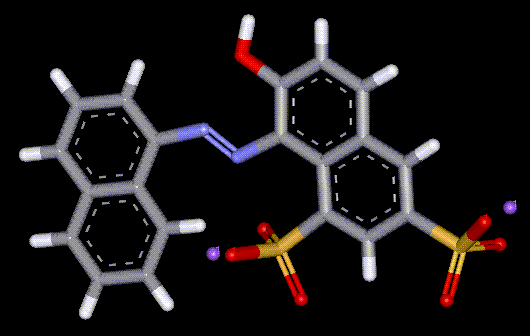 Ponceau 6R is not commonly used, although it has been recommended for staining fibrin with the MSB (Martius, Scarlet and Blue), a reliable trichrome technique.
Ponceau 6R is not commonly used, although it has been recommended for staining fibrin with the MSB (Martius, Scarlet and Blue), a reliable trichrome technique.
| Chemical formula: | C20H12N2O7S2Na2 |
 Erythrosine is a cherry-pink/red synthetic coal tar dye. As well as being used as a food colouring, erythrosine is also used in printing inks, as a biological stain, and for extraction-photometric determination of K, Cd, Pb, Mn, Zn, Ag. It also serves as an adsorption and fluorescent indicator, a dental plaque disclosing agent and a radiopaque medium. It has been suggested that erythrosine may affect thyroid activity due the presence of iodine in the molecule which may be released upon degradation, and that erythrosine may be carcinogenic. The chemical can cause phototoxicity (sensitivity to light).
Erythrosine is a cherry-pink/red synthetic coal tar dye. As well as being used as a food colouring, erythrosine is also used in printing inks, as a biological stain, and for extraction-photometric determination of K, Cd, Pb, Mn, Zn, Ag. It also serves as an adsorption and fluorescent indicator, a dental plaque disclosing agent and a radiopaque medium. It has been suggested that erythrosine may affect thyroid activity due the presence of iodine in the molecule which may be released upon degradation, and that erythrosine may be carcinogenic. The chemical can cause phototoxicity (sensitivity to light).
| Chemical formula: | 2 C20 H6 I4 O5 . 4 Na (Derived). |
| Other names: | 3',6'-Dihydroxy-2',4',5',7'-tetraiodospiro[isobenzofuran-1(3H),9'(9H)-xanthen]-3-one. |
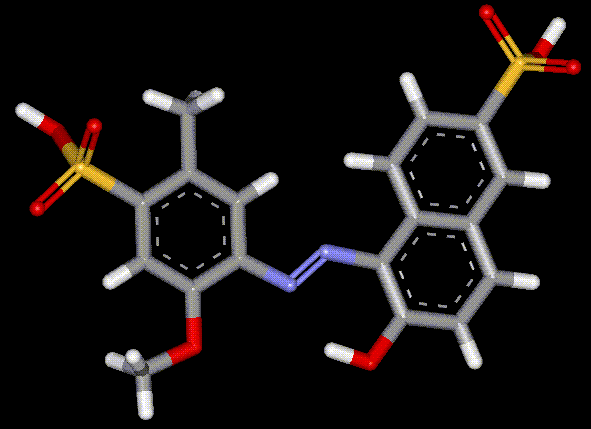 Red 2G is a synthetic red coal tar and azo dye, which is used particularly in meat products. The latter may contain sulfur dioxide and metabisulfite which normally have a bleaching effect, but Red 2G is relatively unaffected. There is evidence that Red 2G can be converted to aniline in the gut. Laboratory tests have shown that Aniline causes anaemia in rats, as it affects haemoglobin in red blood cells.
Red 2G is a synthetic red coal tar and azo dye, which is used particularly in meat products. The latter may contain sulfur dioxide and metabisulfite which normally have a bleaching effect, but Red 2G is relatively unaffected. There is evidence that Red 2G can be converted to aniline in the gut. Laboratory tests have shown that Aniline causes anaemia in rats, as it affects haemoglobin in red blood cells. Allura Re AC is a red synthetic azo dye which was introduced to replace Amaranth. Any allergic reaction to this dye is small compared to reactions to other azo dyes, although asmatics and aspirin intolerant people are still at risk.
Allura Re AC is a red synthetic azo dye which was introduced to replace Amaranth. Any allergic reaction to this dye is small compared to reactions to other azo dyes, although asmatics and aspirin intolerant people are still at risk.| Other names: | 6-Hydroxy-5-[(2-methoxy-5-methyl-4-sulfophenyl)azo]-2-naphthalenesulfonic acid. |
| Appearance: | dark red powder. |
 Indanthrene is a blue dye, the compounds and derivatives of which are used as vat dyestuffs.
Indanthrene is a blue dye, the compounds and derivatives of which are used as vat dyestuffs.| Other names: | 6,15-Dihydro-5,9,14,18-anthrazinetetrone. |
| Chemical formula: | C28 H14 N2 O4 |
| Appearance: | blue needles with metallic lustre. |
| Melting point: | 470 - 500 C |
 Patent Blue V is a synthetic blue-violet coal tar dye used only moderately in the food industry. It is mainly used to colour the lymph vessels, and as a cardiovascular investigative dye. Hypersensitivity reactions reported include itching and nettle rash, nausea, low blood pressure, and in rare cases anaphylactic shock. Patent Blue V is also used as an acid base indicator.
Patent Blue V is a synthetic blue-violet coal tar dye used only moderately in the food industry. It is mainly used to colour the lymph vessels, and as a cardiovascular investigative dye. Hypersensitivity reactions reported include itching and nettle rash, nausea, low blood pressure, and in rare cases anaphylactic shock. Patent Blue V is also used as an acid base indicator.| Other names: | N-[4-[[4-(Diethylamino)phenyl](2,4-disulfophenyl) methylene]-2,5-cyclohexadien-1-ylidene]-N- ethylethanaminium, acid blue V. |
| Chemical formula: | C54H62CAN4O14S4 |
| CAS NO.: | 129-17-9 |
| Appearance: | violet powder. |
| Other information: | Harmful if swallowed, inhaled or absorbed through skin. Causes irritation. |
 Indigo carmine is a blue synthetic coal tar dye, used not only in the food and cosmetics industries but also as a diagnostic aid (e.g. in kidney function tests), as a titrimetric indicator of Cr(II), Sn(II), Fe(III), and Ti(III). Indigo carmine is also used as a photometric detector, and a biological stain. It is normally produced by a synthesis of indoxyl by fusion of sodium phenylglycinate in a mixture of
caustic soda and sodamide. The chemical structure of indigo was determined by Prussian chemist J. F. W. Adolf von Baeyer in 1883.
Indigo carmine is a blue synthetic coal tar dye, used not only in the food and cosmetics industries but also as a diagnostic aid (e.g. in kidney function tests), as a titrimetric indicator of Cr(II), Sn(II), Fe(III), and Ti(III). Indigo carmine is also used as a photometric detector, and a biological stain. It is normally produced by a synthesis of indoxyl by fusion of sodium phenylglycinate in a mixture of
caustic soda and sodamide. The chemical structure of indigo was determined by Prussian chemist J. F. W. Adolf von Baeyer in 1883.| Chemical formula: | C16H8N2Na2O8S2 |
| CAS No: | 860-22-0 |
| Appearance: | dark blue powder |
| Other information: | Indigo Carmine consists essentially of a mixture of disodium 3,3' dioxo - 2,2' -bi-indolylidene-5,5' - disulfonate, disodium 3,3' - dioxo-2,2' - bi-indolyldene-5,7'-disulfonate. Indigo carmine exists as a sodium salt, although the calcium and potassium salts are also permitted. Harmful if swallowed, inhaled or absorbed through the skin. |
 Brilliant blue is a blue synthetic coal tar dye which can be used together with tartrazine to give shades of green. It is also used in inks, as a fabric and wool dye, and to stain proteins.
Brilliant blue is a blue synthetic coal tar dye which can be used together with tartrazine to give shades of green. It is also used in inks, as a fabric and wool dye, and to stain proteins. Commercially available chlorophyll contains other plant pigments, fatty acids and phosphatides, and maybe other oils, fats and waxes. Chlorophyll is an example of a naturally occurring ionophore. It is a tetradentate ligand that binds to an Mg ion through coordination with nitrogen. Chlorophyll is used as a source of olive/dark-green colour, it is susceptible to fading.
Commercially available chlorophyll contains other plant pigments, fatty acids and phosphatides, and maybe other oils, fats and waxes. Chlorophyll is an example of a naturally occurring ionophore. It is a tetradentate ligand that binds to an Mg ion through coordination with nitrogen. Chlorophyll is used as a source of olive/dark-green colour, it is susceptible to fading.  The Mg ion in chlorophyll is substituted by Cu to give a more stable olive green colouring. Chlorophyllins contain other related substituted plant pigments, and provide a water-soluble green colour.
The Mg ion in chlorophyll is substituted by Cu to give a more stable olive green colouring. Chlorophyllins contain other related substituted plant pigments, and provide a water-soluble green colour. Carrots, green leafy vegetables and tomatoes are very good sources of natural carotene which is chemically extracted and is comprised of a mixture of beta-, alpha- and gamma-carotene. Natural carotene, an orange-yellow colouring, contains mainly beta-carotene which has vitamin A as its natural degradation product. Carotene colouring fades on exposure to light.
Carrots, green leafy vegetables and tomatoes are very good sources of natural carotene which is chemically extracted and is comprised of a mixture of beta-, alpha- and gamma-carotene. Natural carotene, an orange-yellow colouring, contains mainly beta-carotene which has vitamin A as its natural degradation product. Carotene colouring fades on exposure to light. Lycopene is a natural red colour derived from tomatoes.
Lycopene is a natural red colour derived from tomatoes. 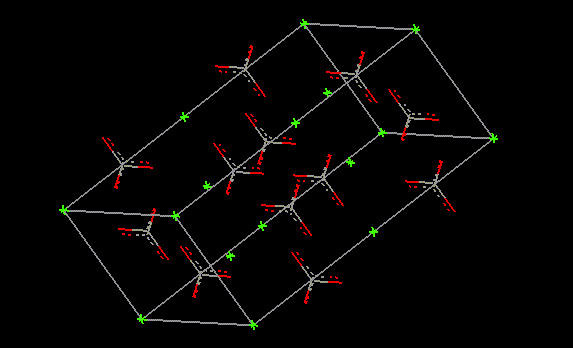 Calcium carbonate occurs naturally and may be used in wine as an acidity regulator, as well as a firming agent for preserved fruit and vegetables. The most common form of calcium carbonate is limestone. Other forms include chalk, marble, corals and calcite. Limestone is a major building material as quick lime (CaO) and slaked lime (Ca(OH)2) are derived from it. Specially precipitated CaCO3 is extensively used in the paper industry as it adds brightness, opacity, ink receptivity and smoothness to the finished product. Calcium carbonate is also used as a filler in rubbers, latex, paints and enamels, and in plastics, as well as finding use as a mild abrasive in toothpastes and an antacid. High levels of ingestion may result in flatulence, constipation, haemmorrhoids and bleeding anal fissures.
Calcium carbonate occurs naturally and may be used in wine as an acidity regulator, as well as a firming agent for preserved fruit and vegetables. The most common form of calcium carbonate is limestone. Other forms include chalk, marble, corals and calcite. Limestone is a major building material as quick lime (CaO) and slaked lime (Ca(OH)2) are derived from it. Specially precipitated CaCO3 is extensively used in the paper industry as it adds brightness, opacity, ink receptivity and smoothness to the finished product. Calcium carbonate is also used as a filler in rubbers, latex, paints and enamels, and in plastics, as well as finding use as a mild abrasive in toothpastes and an antacid. High levels of ingestion may result in flatulence, constipation, haemmorrhoids and bleeding anal fissures. | Other names: | limestone, marble, calcite, chalk |
| Molecular formula: | Ca CO3 |
| CAS No: | 471-34-1 |
| Physical appearance: | white or colourless crystals |
| Melting point: | 825 C |
| Other information: | Dust may cause irritation |
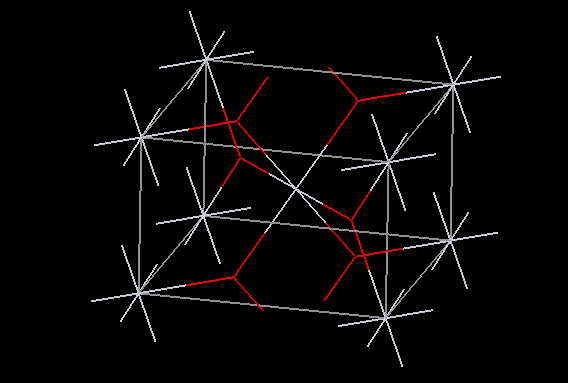 Titanium dioxide may be used in food to give opacity. No adverse effects are known, and the compound is chemically inert. The manufacture of paint constitutes the major use of titanium dioxide, but it is also used as a paper coating and as a filler in rubber and plastics. The most common form of titanium dioxide at room temperature is the rutile structure, which is a slightly distorted hcp of O atoms with half octahedral holes filled by Ti atoms. Fine particles may be used to produce high opacity films as titanium dioxide has an exceptionally high refractive index in the visible region. Naturally occurring forms of titanium dioxide are usually impure, hence the sulphate process or the chloride process are normally used to harvest an acceptable purity of compound.
Titanium dioxide may be used in food to give opacity. No adverse effects are known, and the compound is chemically inert. The manufacture of paint constitutes the major use of titanium dioxide, but it is also used as a paper coating and as a filler in rubber and plastics. The most common form of titanium dioxide at room temperature is the rutile structure, which is a slightly distorted hcp of O atoms with half octahedral holes filled by Ti atoms. Fine particles may be used to produce high opacity films as titanium dioxide has an exceptionally high refractive index in the visible region. Naturally occurring forms of titanium dioxide are usually impure, hence the sulphate process or the chloride process are normally used to harvest an acceptable purity of compound.| Other names: | tiox, unitane, horse head A-420, titan white, numerous trade names |
| Molecular formula: | Ti O2 |
| CAS No: | 1317-70-0 |
| Physical appearance: | white powder |
| Melting point: | 1855 C |
| Other information: | May be harmful or act as an irritant. |
 Alpha-Fe2O3 occurs naturally as the mineral haematite. Together the iron oxides and hydroxides are used to provide red, orange, yellow, brown and black pigments, with the iron not being actively available to body tissues. Iron(III) oxide is also used as a major source of elemental iron in industry. Alpha-iron(III)oxide has the corundum structure.
Alpha-Fe2O3 occurs naturally as the mineral haematite. Together the iron oxides and hydroxides are used to provide red, orange, yellow, brown and black pigments, with the iron not being actively available to body tissues. Iron(III) oxide is also used as a major source of elemental iron in industry. Alpha-iron(III)oxide has the corundum structure.| Other names: | ferric oxide, red iron oxide, iron (2+) oxide |
| Molecular formula: | Fe2 O3 |
| CAS No: | 1309-37-1 |
| Physical appearance: | red to reddish-brown powder |
| Melting point: | 1565 C |
| Other information: | Dust may act as an irritant. |
 Aluminium is a naturally occurring element, present in a normal healthy diet. It is used in food as a metallic surface coating. There is evidence that aluminium accumulation in body cells could be toxic and linked to Parkinson-type diseases, or that skeletal deformations could occur. Pure aluminium is a silvery-white metal that is capable of taking a high polish, has high thermal and electrical conductivity, has excellent corrosion resistance, is non-magnetic, malleable and ductile. It is the most commonly occurring metal in the earth's crust (8.3% by weight), and as well as being used in the food industry, is also used in the construction and aerospace industries, in aerosol cans and foil.
Aluminium is a naturally occurring element, present in a normal healthy diet. It is used in food as a metallic surface coating. There is evidence that aluminium accumulation in body cells could be toxic and linked to Parkinson-type diseases, or that skeletal deformations could occur. Pure aluminium is a silvery-white metal that is capable of taking a high polish, has high thermal and electrical conductivity, has excellent corrosion resistance, is non-magnetic, malleable and ductile. It is the most commonly occurring metal in the earth's crust (8.3% by weight), and as well as being used in the food industry, is also used in the construction and aerospace industries, in aerosol cans and foil. | Other names: | aluminum |
| Molecular formula: | Al |
| CAS No: | 7429-90-5 |
| Physical appearance: | silver foil, shot or powder |
| Melting point: | 660 C |
| Other information: | May be harmful by ingestion or inhalation. |
 Silver is used in food to give a metallic surface colour. However prolonged consumption may lead to argyria, a blue-grey skin. The greatest source of silver now is as a by product in the manufacture of non-ferrous metals such as Cu, Pb, Zn. Silver is usually obtained from crushed silver bearing ore. The actual method of recovery from the ore depends on which metal is predominant in the ore but normally ends by electrolysis using one of two techniques, either the Moebius or Thum Balbach systems. Silver is used extensively in the photographic industry, as well as in silverware and jewellery, electrically, for silvering mirrors and in batteries.
Silver is used in food to give a metallic surface colour. However prolonged consumption may lead to argyria, a blue-grey skin. The greatest source of silver now is as a by product in the manufacture of non-ferrous metals such as Cu, Pb, Zn. Silver is usually obtained from crushed silver bearing ore. The actual method of recovery from the ore depends on which metal is predominant in the ore but normally ends by electrolysis using one of two techniques, either the Moebius or Thum Balbach systems. Silver is used extensively in the photographic industry, as well as in silverware and jewellery, electrically, for silvering mirrors and in batteries.| Other names: | Argentum, shell silver, silver shot |
| Molecular formula: | Ag |
| CAS No: | 7440-22-4 |
| Physical appearance: | lustrous soft white metal |
| Melting point: | 961 C |
| Boiling point: | 2155 C |
| Other information: | Solid silver presents few health hazards. Repeated long-term exposure to silver dust can cause permanent blue-grey staining of eyes, mouth, throat and skin, and may cause eye damage. |
 Gold is used in food to give a metallic surface colour which is very unreactive, however it is also very expensive. Gold is produced by crushing gold-containing rock, the grains of gold are then extracted either by the cyanide process or by amalgamation with mercury. Gold is used to settle international debts, but other uses include manufacture of jewellery, in dentistry, the electronics industry and in the aerospace industry.
Gold is used in food to give a metallic surface colour which is very unreactive, however it is also very expensive. Gold is produced by crushing gold-containing rock, the grains of gold are then extracted either by the cyanide process or by amalgamation with mercury. Gold is used to settle international debts, but other uses include manufacture of jewellery, in dentistry, the electronics industry and in the aerospace industry.| Other names: | gold leaf, gold foil, burnish gold |
| Molecular formula: | Au |
| CAS No: | 7440-57-5 |
| Physical appearance: | yellow metal |
| Melting point: | 1065 C |
| Boiling point: | 2808 C |
| Other information: | Tumorigenic effects have been observed in tests with laboratory animals. |