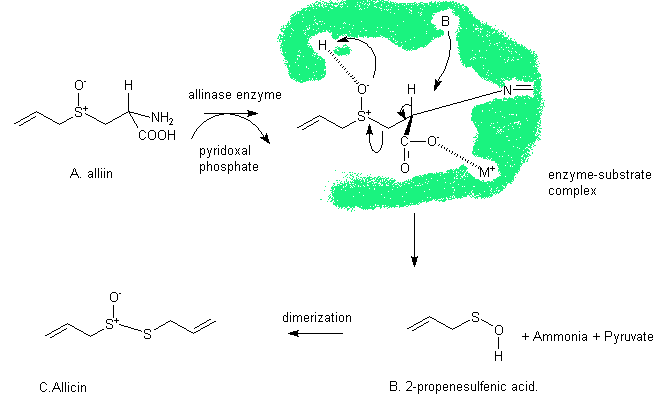
The biochemical reactions that occur in chopped garlic were first probed in 1948 by Stoll and Seebeck of the Sandoz chemical company. They explained why the formation of allicin occurs only on cutting by a enzyme catalysed mechanism. When garlic is chopped or crushed, the garlic tissue is damaged and the precursor, alliin comes into contact with the enzyme allinase. It was later discovered that another biological compound, pyridoxal phosphate, must also be present. The enzyme catalyses the decay of alliin to an unstable intermediate, 2-propenesulfenic acid. This dimerizes to give allicin. Allicin is also unstable and reacts with other compounds present to give many products. More on these products on the next page.
The structure of the lachrymatory precursor in onion is very similiar to that of alliin, so one would expect its reactions to be very similiar. This is true up to a point; the lachrymatory precursor is broken down by allinase to give a 1-propenesulfenic acid. The double bond is in a different place to that formed from garlic, and this double bond allows the sulfenic acid to isomerize to give the lachrymatory factor - the compound that causes tears when chopping onions. This compound has two isomers, syn and anti. Both are formed but the syn compound is formed preferentially. Like allicin it is unstable. It is hydrolysed to give propanal and can dimerize. It can also undergo Diels-Alder cycloaddition with cyclopentadiene.
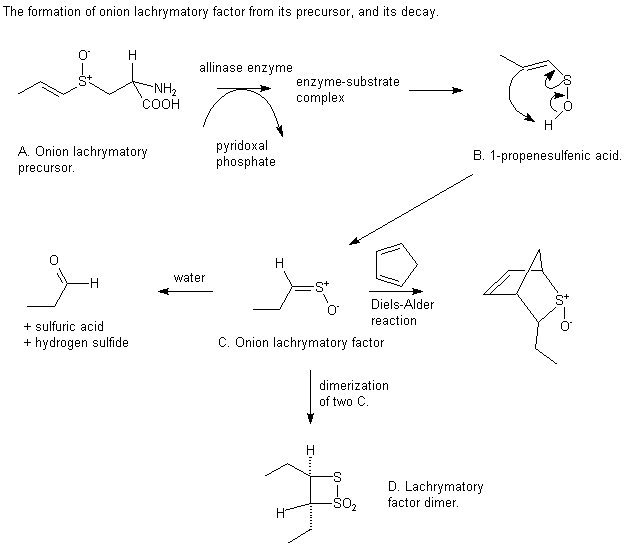
The discomfort sometimes associated with peeling onions can be reduced by two methods. Chilling the onion reduces the volatility of the lachrymatory factor, and peeling under running water dissolves the lachrymatory factor before it can have an effect.
The medicinal properties of garlic have long been known, and have been studied in depth. Continue to the next page for more on the subject.
| introduction | alliums: their curious properties | a little bit of chemistry |
| Medicinal properties and a bit more chemistry | references and further information | links |