


 HCl can be prepared commercially by the reaction of sulphuric acid with sodium chloride, or directly by reaction of hydrogen and chlorine gases; the reaction is very exothermic and takes place readily in sunlight or at elevated temperatures. The major use of HCl is in the manufacture of other chemicals, but other principle applications include metal pickling, gelatine, dyestuffs, casein, pharmaceuticals, synthetic rubber and metal chlorides manufacture. Large amounts of hydrochloric acid are used in effluent treatment and for the regeneration of ion exchange resins in water treatment. Produced in the stomach to aid digestion.
HCl can be prepared commercially by the reaction of sulphuric acid with sodium chloride, or directly by reaction of hydrogen and chlorine gases; the reaction is very exothermic and takes place readily in sunlight or at elevated temperatures. The major use of HCl is in the manufacture of other chemicals, but other principle applications include metal pickling, gelatine, dyestuffs, casein, pharmaceuticals, synthetic rubber and metal chlorides manufacture. Large amounts of hydrochloric acid are used in effluent treatment and for the regeneration of ion exchange resins in water treatment. Produced in the stomach to aid digestion.| Other names: | muriatic acid, chlorohydric acid |
| Molecular formula: | H Cl |
| CAS No: | 7647-01-0 |
| Physical appearance: | clear, colourless or slightly yellow liquid with pungent odour. Concentrated acid is fuming. |
| Melting point: | -25 C |
| Boiling point: | 109 C |
| Other information: | Extremely corrosive. Inhalation of vapour can cause serious injury. Ingestion may be fatal. Liquid can cause severe damage to skin and eyes. |
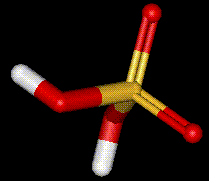 Sulphuric acid was manufactured by the lead-chamber process
Sulphuric acid was manufactured by the lead-chamber process  until the mid-1930s, but this process has now
been replaced by the contact process, involving the catalytic oxidation of sulphur dioxide. The structure of sulphuric acid is considered to be a mixture of several rapidly interconverting equivalent forms.
until the mid-1930s, but this process has now
been replaced by the contact process, involving the catalytic oxidation of sulphur dioxide. The structure of sulphuric acid is considered to be a mixture of several rapidly interconverting equivalent forms.| Other names: | oil of vitriol, mattling acid, vitriol, battery acid |
| Molecular formula: | H2SO4 |
| CAS No: | 7664-93-9 |
| Physical appearance: | Colourless oily liquid |
| Melting point: | -2 C |
| Boiling point: | 327 C |
| Other information: | Extremely corrosive, causes serious burns. Highly toxic. Harmful by inhalation, ingestion and through skin contact. Ingestion may be fatal. Skin contact can lead to extensive and severe burns. Chronic exposure may result in lung damage and possibly cancer. |
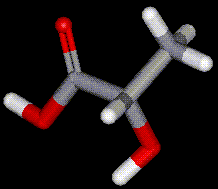 Lactic acid is a naturally occurring compound present in sour milk, molasses and fruit. Lactic acid is produced commercially by fermentation of carbohydrates in the presence of lactic acid bacteria. Lactic acid occurs naturally in the blood (lactates) when glycogen is broken down in the muscles.
Lactic acid is a naturally occurring compound present in sour milk, molasses and fruit. Lactic acid is produced commercially by fermentation of carbohydrates in the presence of lactic acid bacteria. Lactic acid occurs naturally in the blood (lactates) when glycogen is broken down in the muscles.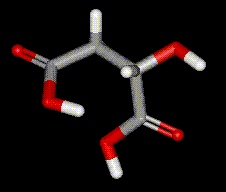 Occurs in two chiral molecules, the D- and L- forms. L-malic acid is a naturally occurring organic acid that is used in the body to derive ATP from food (see the citric acid cycle). It is found in many fruits and vegetables, especially apples. Malic acid may aid in the treatment of fibromyalgia. Commercial malic acid is usually a mixture of the two types, synthesised by heating maleic acid with dilute sulphuric acid, under pressure.
Occurs in two chiral molecules, the D- and L- forms. L-malic acid is a naturally occurring organic acid that is used in the body to derive ATP from food (see the citric acid cycle). It is found in many fruits and vegetables, especially apples. Malic acid may aid in the treatment of fibromyalgia. Commercial malic acid is usually a mixture of the two types, synthesised by heating maleic acid with dilute sulphuric acid, under pressure.| Other names: | DL-Malic acid, hydroxybutanedioic acid |
| Molecular formula: | HOCOCH2CHOHCOOH |
| CAS No: | 617-48-1 |
| Physical appearance: | Odourless white crystals |
| Melting point: | 130 C |
| Boiling point: | 150 C |
| Other information: | Irritant, harmful if swallowed, inhaled or absorbed through skin. |
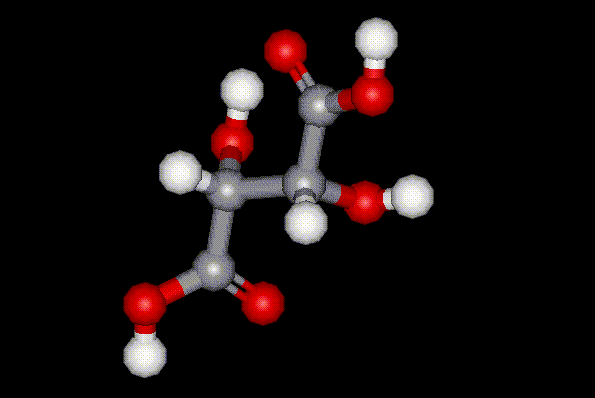 Tartaric acid exists as a pair of enantiomers and an achiral meso compound. (+)-tartaric acid
Tartaric acid exists as a pair of enantiomers and an achiral meso compound. (+)-tartaric acid  commonly occurs in nature and can be found in fruit, and sometimes in wine. Tartaric acid is industrially synthesised as a byproduct during wine making, and it is used in food as an antioxidant and synergist to increase the antioxidant effect of other substances. It is also used as an acidity regulator and sequestrant. Excessive ingestion of tartaric acid results in laxative effects.
commonly occurs in nature and can be found in fruit, and sometimes in wine. Tartaric acid is industrially synthesised as a byproduct during wine making, and it is used in food as an antioxidant and synergist to increase the antioxidant effect of other substances. It is also used as an acidity regulator and sequestrant. Excessive ingestion of tartaric acid results in laxative effects.| Molecular formula: | C4H6O6 |
| CAS No: | 147-71-7 |
| Physical appearance: | white crystals |
| Melting point: | 172 - 174 C |
| Boiling point: | 275 C |
| Other information: | May be harmful by inhalation, ingestion or skin absorption. May act as an irritant. |